05.28.15
The U.S. Food and Drug Administration (FDA) has approved the resumption of patient enrollment in Sunshine Heart Inc.’s its Counter HF US pivotal study for the C-Pulse heart assist system. The company has already begun the process of providing all pivotal study centers with the information required for their Investigational Review Board to approve the continuation of study enrollment.
“Resuming enrollment in Counter HF has been our top priority and was accomplished within the timelines originally announced,” said Dave Rosa, President and CEO of Sunshine Heart. “The proposed protocol modifications have enhanced an already robust protocol, and should increase the likelihood of success. We are encouraged by our investigators’ enthusiasm as witnessed by the high site representation at our recent investigator meeting.”
In February, the FDA unconditionally approved an interim analysis for the Counter HF study as a means to potentially reduce the approval timeline for the C-Pulse therapy. FDA approval to resume study enrollment will now allow Sunshine Heart to progress towards that goal.
Counter HF is a prospective, randomized, multi-center clinical study, and is being conducted by heart failure and cardiac surgeon specialists in the United States. The trial is expected to randomize 388 patients in up to 40 clinical sites. The purpose of the study is to determine whether the C-Pulse system is a safe and effective treatment for heart failure patients who meet certain levels of heart failure criteria.
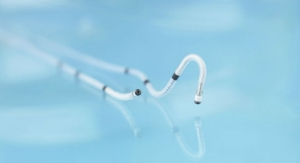
The C-Pulse heart assist system, or C-Pulse system, an investigational device in the United States, Canada and countries that do not recognize the CE mark approval, uses the scientific principles of intra-aortic balloon counter-pulsation applied in an extra-aortic approach to assist the left ventricle by reducing the workload required to pump blood throughout the body, while increasing blood flow to the coronary arteries. Combined, these potential benefits may help sustain the patient’s current condition or, in some cases, reverse the heart failure process, thereby potentially preventing the need for later-stage heart failure devices, such as left ventricular assist devices (LVADs), artificial hearts or transplants. It may also provide relief from the symptoms of class III and ambulatory class IV heart failure and improve quality of life and cardiac function.
“Resuming enrollment in Counter HF has been our top priority and was accomplished within the timelines originally announced,” said Dave Rosa, President and CEO of Sunshine Heart. “The proposed protocol modifications have enhanced an already robust protocol, and should increase the likelihood of success. We are encouraged by our investigators’ enthusiasm as witnessed by the high site representation at our recent investigator meeting.”
In February, the FDA unconditionally approved an interim analysis for the Counter HF study as a means to potentially reduce the approval timeline for the C-Pulse therapy. FDA approval to resume study enrollment will now allow Sunshine Heart to progress towards that goal.
Counter HF is a prospective, randomized, multi-center clinical study, and is being conducted by heart failure and cardiac surgeon specialists in the United States. The trial is expected to randomize 388 patients in up to 40 clinical sites. The purpose of the study is to determine whether the C-Pulse system is a safe and effective treatment for heart failure patients who meet certain levels of heart failure criteria.
The C-Pulse heart assist system, or C-Pulse system, an investigational device in the United States, Canada and countries that do not recognize the CE mark approval, uses the scientific principles of intra-aortic balloon counter-pulsation applied in an extra-aortic approach to assist the left ventricle by reducing the workload required to pump blood throughout the body, while increasing blood flow to the coronary arteries. Combined, these potential benefits may help sustain the patient’s current condition or, in some cases, reverse the heart failure process, thereby potentially preventing the need for later-stage heart failure devices, such as left ventricular assist devices (LVADs), artificial hearts or transplants. It may also provide relief from the symptoms of class III and ambulatory class IV heart failure and improve quality of life and cardiac function.