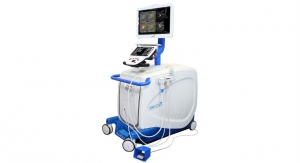
Koning Corporation has received U.S. Food and Drug Administration (FDA) approval for its 3-D breast CT scanner, including a biopsy bracket add-on.
Koning completed the FDA's premarket approval (PMA) process for its Koning Breast computed tomography (CT) system, as well as an optional biopsy bracket for CT-guided biopsies of suspicious breast lesions. Other optional accessories for the system include a collimator that limits the x-ray beam to the area of interest, according to the vendor.
"This FDA approval represents a major step forward for breast imaging and women's health care," said Ruola Ning, Ph.D., Koning's president/founder and sole inventor of cone beam breast CT technology. "KBCT represents an advancement in breast cancer diagnosis. Breast cancer is a growing worldwide women's health issue impacting hundreds of thousands of women. We are very proud to now be able to offer our technology to benefit women here in the United States."
KBCT is the first commercially available, 3-D breast CT scanner designed specifically to image the entire breast with a single scan without compressing the breast tissue. The system acquires hundreds of images in 10 seconds, producing 'true' 3-D images to expedite procedures and minimize patient discomfort. Optional accessories for KBCT include a biopsy bracket and a collimator which is used to limit the X-ray beam to the targeted area. The biopsy bracket provides 3-D targeting at comparable or lower radiation exposure compared to stereotactic guided biopsy, the company said.
In a September 2014 FDA Consumer Health Information update ("3D Technologies Poised to Change How Doctors Diagnose Cancer") FDA reported that current advances in 3-D imaging would soon help doctors find hidden tumors and better diagnose cancer. The article indicated that breast CT procedures are more comfortable than regular mammograms because the breast is not compressed. There also is less radiation exposure during breast CT procedures than during CT chest exams because only the breast is exposed to X-rays. In addition, the update said, breast CT images have less distortion than mammography, and the system is optimized to differentiate between the breast's soft tissue and cancer tissue. The images allow clinicians to view the breast from any orientation (up and down, left and right) and get a unique view of the breast --- almost like seeing the anatomy itself.
More than 680 patient scans on KBCT were conducted at Elizabeth Wende Breast Care (Rochester, N.Y.) and the University of Rochester Medical Center (URMC-Rochester, N.Y.), with additional collaboration at the University of Massachusetts Medical Center (Wooster, Mass.) which culminated in a large reader study conducted at the Medical College of South Carolina with Etta Pisano, M.D., a world renowned expert in breast imaging, serving as the principal investigator.
"The results we have seen with 3D-KBCT have been remarkable compared to 2-D imaging and there is no compression of the tissue making breast CT a much more comfortable and painless procedure for women. I believe that 3D-KBCT will likely play a major role for multiple applications in breast imaging," Pisano said.
Koning Corporation is a West Henrietta, N.Y.-based medical imaging company that is developing and marketing cone beam CT scanners. KBCT has been approved for sale in Canada, Australia, and the European Union (CE Mark). Koning anticipates Chinese approval soon.