05.21.15
Lumicell, a developer of technology for image-guided cancer surgery, has received investigational device exemption approval from the U.S. Food and Drug Administration (FDA) to launch a feasibility study for intraoperative imaging of breast cancer during surgery. The study will be conducted at Massachusetts General Hospital (MGH) in Boston.
The National Cancer Institute (NCI) has funded Lumicell’s pre-clinical toxicology tests and the manufacturing of Lumicell’s imaging agent for the pilot clinical trials through the NCI Experimental Therapeutics (NExT) program. In addition, in collaboration with the director of the Breast Cancer Program at MGH, Lumicell helped secure additional NCI funding from an R21 Early Phase Trials program to support the feasibility study in breast cancer.
Lumicell recently completed a Phase 1 safety study with its leading imaging agent LUM015 at Duke University Medical Center in Durham, N.C., in the indications of breast cancer and sarcoma with no agent-related adverse events observed.
The principal investigator for the feasibility study is Barbara Smith, M.D., director, Breast Cancer Program and co-director, Gillette Center for Women’s Cancer at MGH.
“Launching a feasibility study for intraoperative imaging of breast cancer during surgery is a critical next step for Lumicell,” said W. David Lee, CEO of Lumicell. “The NCI grant allows our team to demonstrate the efficacy of our system in reducing rates of positive margins and repeat surgeries. By enabling surgeons to remove sub-millimeter residual cancer, we expect to improve patient outcomes.
“The NCI is the nation’s leader in cancer research and their support of our work has been critical as we move our technology forward,” added Lee. “NCI’s funding of our pre-clinical toxicity studies and the manufacturing of our imaging agent, LUM015, for the feasibility study through the NCI NExT program, has helped drive our early successes.”
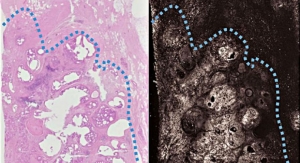
According to its leadership, Lumicell is addressing the “number one challenge in cancer surgery,” which is the need to remove all cancer cells within the tumor bed during the first surgery. In the field of cancer surgery, the goal is clean margins. Lumicell’s intraoperative detection system provides surgeons with the tools to perform a thorough removal of any residual cancer cells in real-time within the tumor bed thus eliminating the need for repeat surgeries due to positive margins or local recurrence, company officials claim.
Lumicell’s surgical system includes a lightweight, hand-held single-cell detection imaging device and a cancer-specific molecular imaging agent. This image-guided technology is designed to fit within the existing surgical workflow. The company’s devices and imaging agents are optimized for the specific cancer-type and surgical procedure.
Wellesley, Mass.-based Lumicell initially is investigating its imaging system in patients undergoing surgery for breast cancer and sarcomas. Additional future indications will include surgeries for lung, prostate, ovarian, colorectal and brain cancers.
The National Cancer Institute (NCI) has funded Lumicell’s pre-clinical toxicology tests and the manufacturing of Lumicell’s imaging agent for the pilot clinical trials through the NCI Experimental Therapeutics (NExT) program. In addition, in collaboration with the director of the Breast Cancer Program at MGH, Lumicell helped secure additional NCI funding from an R21 Early Phase Trials program to support the feasibility study in breast cancer.
Lumicell recently completed a Phase 1 safety study with its leading imaging agent LUM015 at Duke University Medical Center in Durham, N.C., in the indications of breast cancer and sarcoma with no agent-related adverse events observed.
The principal investigator for the feasibility study is Barbara Smith, M.D., director, Breast Cancer Program and co-director, Gillette Center for Women’s Cancer at MGH.
“Launching a feasibility study for intraoperative imaging of breast cancer during surgery is a critical next step for Lumicell,” said W. David Lee, CEO of Lumicell. “The NCI grant allows our team to demonstrate the efficacy of our system in reducing rates of positive margins and repeat surgeries. By enabling surgeons to remove sub-millimeter residual cancer, we expect to improve patient outcomes.
“The NCI is the nation’s leader in cancer research and their support of our work has been critical as we move our technology forward,” added Lee. “NCI’s funding of our pre-clinical toxicity studies and the manufacturing of our imaging agent, LUM015, for the feasibility study through the NCI NExT program, has helped drive our early successes.”
According to its leadership, Lumicell is addressing the “number one challenge in cancer surgery,” which is the need to remove all cancer cells within the tumor bed during the first surgery. In the field of cancer surgery, the goal is clean margins. Lumicell’s intraoperative detection system provides surgeons with the tools to perform a thorough removal of any residual cancer cells in real-time within the tumor bed thus eliminating the need for repeat surgeries due to positive margins or local recurrence, company officials claim.
Lumicell’s surgical system includes a lightweight, hand-held single-cell detection imaging device and a cancer-specific molecular imaging agent. This image-guided technology is designed to fit within the existing surgical workflow. The company’s devices and imaging agents are optimized for the specific cancer-type and surgical procedure.
Wellesley, Mass.-based Lumicell initially is investigating its imaging system in patients undergoing surgery for breast cancer and sarcomas. Additional future indications will include surgeries for lung, prostate, ovarian, colorectal and brain cancers.