05.19.15
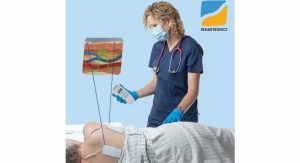
BrainScope Company Inc. received U.S. Food and Drug Administration (FDA) clearance for its Ahead 200 device. Using commercial smartphone hardware that leverages Google’s Android operating system, the Ahead 200 records and analyzes a patient’s electroencephalograph (EEG) using a custom sensor attached to the handheld to provide an interpretation of the structural condition of the patient’s brain after head injury.
In late 2012, BrainScope announced that it had been awarded a $2.67 million contract by the United States Army for development of the Ahead 200, a smaller, more rugged and modernized version of its Ahead 100 traumatic brain injury assessment technology. Similar to the Ahead 100, which was cleared this past November, it is indicated for use as an adjunct to standard clinical practice to aid in the evaluation of patients who are being considered for a head computerized tomography (CT) scan, but should not be used as a substitute for a CT scan. It is to be used on patients who sustained a closed head injury within 24 hours, clinically present as a mild traumatic brain injury (TBI), and are between the ages of 18-80 years.
“We are particularly grateful for the strong, continuing partnership with the [U.S.] Department of Defense,” said Michael Singer, president and CEO of Bethesda, Md.-based BrainScope. “FDA clearance of our Ahead 200 represents a significant achievement in BrainScope’s mission to develop an objective, non-invasive, patient-friendly assessment device for rapid and easy use in urgent care settings. A device with the ability to rapidly identify and categorize patients who present with mild symptoms but who may have a life-threatening TBI has the potential to improve triage, save lives, reduce radiation exposure and decrease costs to the healthcare system.”
In September 2014, BrainScope announced that it had been awarded three contracts valued at $15.9 million by the U.S. Department of Defense for continued research and development of the Ahead system. The contracts support enhancement of BrainScope’s Ahead technology and current nationwide, multi-site clinical studies in hospital emergency rooms, and extend its TBI focus to concussion assessment technology. Studies in concussed athletes are underway in cooperation with university sports programs. These activities leverage six years of BrainScope studies in sports venues, including a study awarded through the GE-NFL Head Health Challenge I.
“The Ahead 200 project in collaboration with the U.S. Department of Defense has culminated in another important FDA clearance and has allowed us to leverage the ubiquitous Android mobile operating system and advances in handheld processing technology fueled by the smartphone market to develop and ultimately commercialize our products. We continue to refine the ultimate product through our ongoing clinical studies and technology development,” Singer said.
Results from independent clinical studies using BrainScope’s technology have been published through 15 articles in leading peer-reviewed neurotrauma and emergency medicine journals such as Journal of Neurotrauma, Brain Injury, Academic Emergency Medicine, Journal of Head Trauma Rehabilitation, The American Journal of Emergency Medicine, and Military Medicine.
BrainScope holds 86 issued and pending patents related to its technology.
BrainScope has been awarded more than $27 million of U.S. Department of Defense research contracts for the development of its TBI assessment technology and has received significant funding from private investors to accelerate its development efforts.
In late 2012, BrainScope announced that it had been awarded a $2.67 million contract by the United States Army for development of the Ahead 200, a smaller, more rugged and modernized version of its Ahead 100 traumatic brain injury assessment technology. Similar to the Ahead 100, which was cleared this past November, it is indicated for use as an adjunct to standard clinical practice to aid in the evaluation of patients who are being considered for a head computerized tomography (CT) scan, but should not be used as a substitute for a CT scan. It is to be used on patients who sustained a closed head injury within 24 hours, clinically present as a mild traumatic brain injury (TBI), and are between the ages of 18-80 years.
“We are particularly grateful for the strong, continuing partnership with the [U.S.] Department of Defense,” said Michael Singer, president and CEO of Bethesda, Md.-based BrainScope. “FDA clearance of our Ahead 200 represents a significant achievement in BrainScope’s mission to develop an objective, non-invasive, patient-friendly assessment device for rapid and easy use in urgent care settings. A device with the ability to rapidly identify and categorize patients who present with mild symptoms but who may have a life-threatening TBI has the potential to improve triage, save lives, reduce radiation exposure and decrease costs to the healthcare system.”
In September 2014, BrainScope announced that it had been awarded three contracts valued at $15.9 million by the U.S. Department of Defense for continued research and development of the Ahead system. The contracts support enhancement of BrainScope’s Ahead technology and current nationwide, multi-site clinical studies in hospital emergency rooms, and extend its TBI focus to concussion assessment technology. Studies in concussed athletes are underway in cooperation with university sports programs. These activities leverage six years of BrainScope studies in sports venues, including a study awarded through the GE-NFL Head Health Challenge I.
“The Ahead 200 project in collaboration with the U.S. Department of Defense has culminated in another important FDA clearance and has allowed us to leverage the ubiquitous Android mobile operating system and advances in handheld processing technology fueled by the smartphone market to develop and ultimately commercialize our products. We continue to refine the ultimate product through our ongoing clinical studies and technology development,” Singer said.
Results from independent clinical studies using BrainScope’s technology have been published through 15 articles in leading peer-reviewed neurotrauma and emergency medicine journals such as Journal of Neurotrauma, Brain Injury, Academic Emergency Medicine, Journal of Head Trauma Rehabilitation, The American Journal of Emergency Medicine, and Military Medicine.
BrainScope holds 86 issued and pending patents related to its technology.
BrainScope has been awarded more than $27 million of U.S. Department of Defense research contracts for the development of its TBI assessment technology and has received significant funding from private investors to accelerate its development efforts.