04.23.15
The National University Hospital, Singapore (NUH), and ALung Technologies, developer of low-flow extracorporeal CO2 removal (ECCO2R) technologies for treating patients with acute respiratory failure, have together initiated a new clinical study of the Hemolung Respiratory Assist System (RAS) to provide ECCO2R in patients with moderate-to-severe acute respiratory distress syndrome (ARDS). The first patient was recently enrolled in the study.
ARDS affects the critically ill, and is typically triggered by pneumonia, sepsis or trauma. These patients often require mechanical ventilation as a life saving measure. Mechanical ventilation can sometimes cause further lung injury, known as ventilator induced lung injury (VILI), which is thought to be the primary mechanism of worsening ARDS. This creates a difficult clinical situation, where the patient needs the ventilator to survive, but the ventilator can make the ARDS worse, resulting in mortality rates of up to 50 percent. A promising technique to break this vicious cycle of lung injury is the use of smaller ventilation volumes to achieve ultra-protective ventilation. However, when ventilation is reduced to protect the lung, carbon dioxide quickly accumulates in the patient’s blood, which causes the blood to become very acidic, and can impair the function of the heart and the immune system.
In this pilot study, Matthew Cove, M.D., intensivist and consultant, Division of Respiratory and Critical Care Medicine at the NUH, and his colleagues Zudin Puthucheary, M.D., Ph.D., and Jason Phua MBBS, MRCP, will assess the ability of low-flow ECCO2R provided by the Hemolung RAS to facilitate an ultra-protective ventilation bundle of care. The primary aim of this study is to demonstrate that this bundle of care can be safely applied while avoiding the negative effects of high carbon dioxide levels (hypercapnia) which result when ventilation volumes are reduced.
“To date, only one major intervention has consistently shown reduced mortality in ARDS patients—low tidal volume ventilation, referred to as lung protective ventilation,” said Cove. “Despite these advancements, the optimal volume which protects the injured lung is not yet known. However, mounting evidence suggests that lower tidal volumes and plateau pressures than those we currently use, a strategy called ultra-protective ventilation, may be even more effective. A side effect of this strategy is carbon dioxide retention, which in this study will be controlled through the provision of ECCO2R with the Hemolung RAS.”
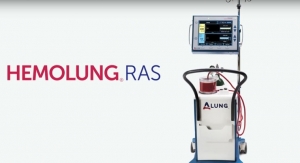
Prior studies of ECCO2R in ARDS patients have produced positive outcomes, but were limited in part by the capabilities of the previously available devices. Cove noted, “With the Hemolung device, we can now perform extracorporeal CO2 removal with a single venous catheter, which is similar to a dialysis catheter, and flow rates similar to those used in kidney dialysis. Our goal is to halt the progression of ARDS and facilitate lung recovery, while avoiding more invasive treatments like extracorporeal membrane oxygenation.”
“Dr. Cove and his team have developed a unique protocol which combines what we believe are all of the latest and most clinically effective strategies for improving outcomes in this group of ARDS patients for which mortality remains very high,” said Peter DeComo, chairman and CEO of ALung. “We are very excited to support this study and anxiously look forward to its results.”
ALung Technologies Inc. is a Pittsburgh, Pa.-based developer and manufacturer of lung assist devices.
ARDS affects the critically ill, and is typically triggered by pneumonia, sepsis or trauma. These patients often require mechanical ventilation as a life saving measure. Mechanical ventilation can sometimes cause further lung injury, known as ventilator induced lung injury (VILI), which is thought to be the primary mechanism of worsening ARDS. This creates a difficult clinical situation, where the patient needs the ventilator to survive, but the ventilator can make the ARDS worse, resulting in mortality rates of up to 50 percent. A promising technique to break this vicious cycle of lung injury is the use of smaller ventilation volumes to achieve ultra-protective ventilation. However, when ventilation is reduced to protect the lung, carbon dioxide quickly accumulates in the patient’s blood, which causes the blood to become very acidic, and can impair the function of the heart and the immune system.
In this pilot study, Matthew Cove, M.D., intensivist and consultant, Division of Respiratory and Critical Care Medicine at the NUH, and his colleagues Zudin Puthucheary, M.D., Ph.D., and Jason Phua MBBS, MRCP, will assess the ability of low-flow ECCO2R provided by the Hemolung RAS to facilitate an ultra-protective ventilation bundle of care. The primary aim of this study is to demonstrate that this bundle of care can be safely applied while avoiding the negative effects of high carbon dioxide levels (hypercapnia) which result when ventilation volumes are reduced.
“To date, only one major intervention has consistently shown reduced mortality in ARDS patients—low tidal volume ventilation, referred to as lung protective ventilation,” said Cove. “Despite these advancements, the optimal volume which protects the injured lung is not yet known. However, mounting evidence suggests that lower tidal volumes and plateau pressures than those we currently use, a strategy called ultra-protective ventilation, may be even more effective. A side effect of this strategy is carbon dioxide retention, which in this study will be controlled through the provision of ECCO2R with the Hemolung RAS.”
Prior studies of ECCO2R in ARDS patients have produced positive outcomes, but were limited in part by the capabilities of the previously available devices. Cove noted, “With the Hemolung device, we can now perform extracorporeal CO2 removal with a single venous catheter, which is similar to a dialysis catheter, and flow rates similar to those used in kidney dialysis. Our goal is to halt the progression of ARDS and facilitate lung recovery, while avoiding more invasive treatments like extracorporeal membrane oxygenation.”
“Dr. Cove and his team have developed a unique protocol which combines what we believe are all of the latest and most clinically effective strategies for improving outcomes in this group of ARDS patients for which mortality remains very high,” said Peter DeComo, chairman and CEO of ALung. “We are very excited to support this study and anxiously look forward to its results.”
ALung Technologies Inc. is a Pittsburgh, Pa.-based developer and manufacturer of lung assist devices.