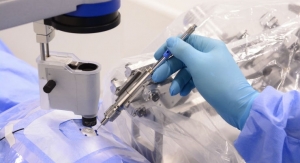
A surgical team coordinated by U.S. Medical Innovations LLC (USMI) recently completed the world’s first clinical procedure using cold plasma for the selective killing of cancerous tissue.
The surgery was performed at the Baton Rouge General Medical Center in Baton Rouge, La., on a patient who previously had been diagnosed with inoperable stage 4 colon cancer. The procedure was cleared through a one-time pre-market compassionate use exemption from the U.S. Food and Drug Administration (FDA). The new cold plasma device used in the procedure has not yet been cleared by the FDA for general commercial use or marketing. The company plans to submit an application for such authorization during the coming months.
According to the company, the surgery was the first of its kind in the world, using its Canady Helios Cold Plasma Scalpel, a new technology that specifically targets cancer cells while sparing normal cells and tissue. The scalpel is a cold atmospheric plasma device used to eradicate microscopic remnants of the cancer, which is used in conjunction with the Canady Hybrid Plasma Scalpel, a device approved and in use since 2011.
USMI CEO, Chief Science Officer and surgeon, Jerome Canady, M.D., designed the clinical and treatment protocols, and guided the surgical team through the procedure. A multidisciplinary group of healthcare professionals, physicians, engineers and scientists collaborated during a 12-month period leading up to the procedure. Extensive treatment plans were implemented including preoperative chemotherapy and radiation therapies to shrink the tumor. Computed tomography scans were used to develop the 3-D computer guided imagery to map out the surgical procedures for the patient, who was initially diagnosed as surgically inoperable.
Stephen V. Gordon, M.D., of the Baton Rouge General Medical Center, completed the surgical procedure under the watch of Canady. The guidance of several top consulting scientists to the USMI family of companies was leveraged in this work as well, including input from Michael Keidar, Ph.D., the co-inventor of the technology, who is also a professor in the Department of Mechanical and Aerospace Engineering at The George Washington University in Washington, D.C.
“For years we’ve worked tirelessly in a global race against time to leverage innovative plasma technologies in the pursuit of the cure for cancer. As other research teams based in Germany, Japan, Korea and elsewhere continue to pursue this research in the lab, we are proud to be the first to utilize cold plasma technology in a surgical device within a clinical setting to prolong the life of this remarkable patient,” said Canady.
The procedure was cleared by the Food and Drug Administration as an emergency measure, after receipt of the application and request from Canady Biotechnology LLC, manufacturer of the USMI surgical product line. The devices used in the procedure were designed by another USMI company—Plasma Medicine Life Sciences LLC, while the translational and molecular protocols were developed by the Jerome Canady Research Institute for Advanced Biological and Technological Sciences, Benefit LLC, a research organization also within the USMI family of companies. ]
Takoma Park, Md.-based USMI makes plasma technology for the eradication of cancer. The company’s products are designed for ambulatory and inpatient endoscopy centers as well as complex surgical procedures.