04.09.15
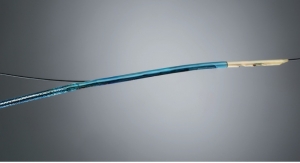
Roxwood Medical Inc. has been granted U.S. Food and Drug Administration (FDA) clearance of its CenterCross catheter for use in the coronary and peripheral vasculature.
The CenterCross Catheter is designed to be used in conjunction with guidewires and microcatheters to access discrete regions of the coronary and peripheral vasculature and enable clinicians to better address wire-crossing of complex lesions. The catheter incorporates a unique self-expanding scaffold to stabilize off-the-shelf interventional tools, such as guidewires and microcatheters, in the center of the artery near the target lesion.
“We’re thrilled with the initial reception that our CenterCross Catheter is receiving clinically. The clearance by the FDA of our second product represents an important milestone for the company and allows us to work with a broad group of interventionalists across the country," said Roxwood Medical President/CEO Mehrdad Farhangnia.
Initial cases using the CenterCross Catheter were performed by some various interventional cardiologists across the United States, including Jihad Mustapha, M.D., of Metro Heart Hospital in Wyoming, Mich., for peripheral cases and Emmanuoil Brilakis, M.D., at the Veteran’s Administration Hospital in Dallas, Texas, for coronary cases.
“My initial cases with the CenterCross Catheter were impressive and consistent with my prior experience with MultiCross,” said Mustapha. “The CenterCross device tracked nicely through torturous anatomy and offers a surprising level of support, allowing me to cross tough lesions more effectively. These tend to be the most challenging and time-consuming procedures, so I’m really excited about the benefits CenterCross offers to simplify and shorten these cases, especially for patients dealing with Critical Limb Ischemia.”
“CenterCross offers an exciting new platform to tackle some of the most complex cases we face in treating disease in the coronaries,” added Brilakis. “It provides excellent support and centering of my coronary support catheter to allow for luminal guidewire crossing. This is significant for patients who need coronary revascularization, but often have limited treatment options.”
Based in Redwood City, Calif., Roxwood Medical is a privately held company that develops catheter platforms for cardiovascular disease. The US FDA has granted the company 510(k) clearance for use of the MultiCross and the CenterCross catheters.
The CenterCross Catheter is designed to be used in conjunction with guidewires and microcatheters to access discrete regions of the coronary and peripheral vasculature and enable clinicians to better address wire-crossing of complex lesions. The catheter incorporates a unique self-expanding scaffold to stabilize off-the-shelf interventional tools, such as guidewires and microcatheters, in the center of the artery near the target lesion.
“We’re thrilled with the initial reception that our CenterCross Catheter is receiving clinically. The clearance by the FDA of our second product represents an important milestone for the company and allows us to work with a broad group of interventionalists across the country," said Roxwood Medical President/CEO Mehrdad Farhangnia.
Initial cases using the CenterCross Catheter were performed by some various interventional cardiologists across the United States, including Jihad Mustapha, M.D., of Metro Heart Hospital in Wyoming, Mich., for peripheral cases and Emmanuoil Brilakis, M.D., at the Veteran’s Administration Hospital in Dallas, Texas, for coronary cases.
“My initial cases with the CenterCross Catheter were impressive and consistent with my prior experience with MultiCross,” said Mustapha. “The CenterCross device tracked nicely through torturous anatomy and offers a surprising level of support, allowing me to cross tough lesions more effectively. These tend to be the most challenging and time-consuming procedures, so I’m really excited about the benefits CenterCross offers to simplify and shorten these cases, especially for patients dealing with Critical Limb Ischemia.”
“CenterCross offers an exciting new platform to tackle some of the most complex cases we face in treating disease in the coronaries,” added Brilakis. “It provides excellent support and centering of my coronary support catheter to allow for luminal guidewire crossing. This is significant for patients who need coronary revascularization, but often have limited treatment options.”
Based in Redwood City, Calif., Roxwood Medical is a privately held company that develops catheter platforms for cardiovascular disease. The US FDA has granted the company 510(k) clearance for use of the MultiCross and the CenterCross catheters.