01.27.15
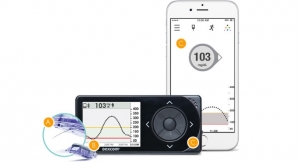
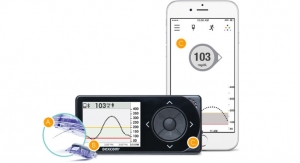
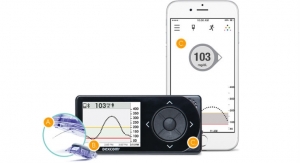
Just over two months after approving the software, the U.S. Food and Drug Administration (FDA) has approved Dexcom Inc.’s G4 Platinum continuous glucose monitoring system with share. The Dexcom Share receiver uses a secure wireless connection via bluetooth low energy (BLE) between a patient’s receiver and an app on the patient’s smartphone to transmit glucose information to apps on the mobile devices of up to five designated recipients, or “followers,” without the need for a dedicated docking cradle. These followers can remotely monitor a patient’s glucose information and receive alert notifications from almost anywhere, initially via their Apple iPhone or iPod touch and in the future on Android devices, giving them peace of mind and reassurance when they are apart. The “Share” and “Follower” apps will be available on the Apple online app store at no charge.
“The Dexcom Share receiver represents a significant step forward for our company and our mobile strategy, but more importantly, it will provide a huge improvement for people managing their diabetes and for those parents and caregivers who help them each and every day,” said Kevin Sayer, president and CEO of Dexcom. “The FDA understands the importance of this type of innovation and the need to regulate it appropriately, and we could not be more pleased with the speed at which they reviewed and approved this important innovation.”
The Share receiver is anticipated to ship to new patients in early March 2015. All patients who purchased a Share cradle will receive a free upgrade to the Share receiver, the company promises; as will all purchasers of a G4 Platinum receiver from January 1, 2015, until the Share receiver is shipped, will receive a free upgrade to the Share receiver. There will be a low cost cash upgrade to the Share receiver for those patients who are still under warranty with their existing receiver. The Share receiver will be compatible with future generation Dexcom sensor systems.
Headquartered in San Diego, Calif., Dexcom makes continuous glucose monitoring systems for ambulatory use by people with diabetes and by healthcare providers in the hospital.
“The Dexcom Share receiver represents a significant step forward for our company and our mobile strategy, but more importantly, it will provide a huge improvement for people managing their diabetes and for those parents and caregivers who help them each and every day,” said Kevin Sayer, president and CEO of Dexcom. “The FDA understands the importance of this type of innovation and the need to regulate it appropriately, and we could not be more pleased with the speed at which they reviewed and approved this important innovation.”
The Share receiver is anticipated to ship to new patients in early March 2015. All patients who purchased a Share cradle will receive a free upgrade to the Share receiver, the company promises; as will all purchasers of a G4 Platinum receiver from January 1, 2015, until the Share receiver is shipped, will receive a free upgrade to the Share receiver. There will be a low cost cash upgrade to the Share receiver for those patients who are still under warranty with their existing receiver. The Share receiver will be compatible with future generation Dexcom sensor systems.
Headquartered in San Diego, Calif., Dexcom makes continuous glucose monitoring systems for ambulatory use by people with diabetes and by healthcare providers in the hospital.