01.09.15
Minneapolis, Minn.-based Medtronic Inc. has begun a randomized clinical trial in Europe to assess the benefits of ablation using the Medtronic Artic Front Advance cryoballoon as a first-line treatment for atrial fibrillation (AF) patients. While medication is currently considered first-line treatment for AF, clinical research indicates that half of all patients with symptomatic disease do not respond to drug therapy; earlier ablation may improve treatment effectiveness.
The Cryo-FIRST (Catheter Cryoablation Versus Antiarrhythmic Drug as First-Line Therapy of Paroxysmal Atrial Fibrillation) trial will evaluate the procedural success and clinical outcomes of patients with paroxysmal AF (PAF) undergoing cryoballoon ablation as an initial treatment compared with medication. It will include approximately 218 patients in up to 12 centers across Europe.
The most recent European guidelines highlight that ablation should be considered as first-line therapy in selected patients with symptomatic PAF. Cryoballoon ablation is not approved as a first-line treatment in the United States.
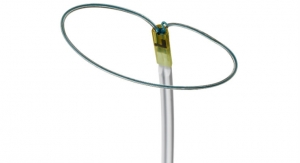
Approximately 7 million people worldwide currently suffer from AF, an irregular quivering or rapid heart rhythm in the upper chambers of the heart that is associated with increased morbidity and mortality as well as added healthcare costs. Cryoablation involves a minimally invasive procedure that creates lesions by freezing tissue in the heart`s upper chambers, traditionally around the pulmonary veins, to block the electrical signals that trigger erratic heart rhythms. Pulmonary vein isolation (PVI) is a standard treatment for patients in the early stages of AF when the triggers come largely from these veins. However, as the disease progresses, it becomes more complicated to treat, with lower long-term success rates.
“For select patients with symptomatic paroxysmal AF and minimal or no heart disease, this therapy—with its established safety profile—has potential as a first-line treatment, providing valuable clinical insights and evidence for future therapeutic solutions,” said Heinz-Friedrich Pitschner, M.D. of the Kerckhoff-Klinik Heart Center in Germany and chair of the Cryo-FIRST steering committee.
“With the prevalence of AF expected to increase exponentially over the next generation, we believe that cryoballoon ablation therapy may safely and effectively address the disease before it progresses, improving outcomes and quality of life for patients, and reducing healthcare costs,” said Reggie Groves, vice president and general manager of Medtronic’s AF Solutions business.
The Cryo-FIRST (Catheter Cryoablation Versus Antiarrhythmic Drug as First-Line Therapy of Paroxysmal Atrial Fibrillation) trial will evaluate the procedural success and clinical outcomes of patients with paroxysmal AF (PAF) undergoing cryoballoon ablation as an initial treatment compared with medication. It will include approximately 218 patients in up to 12 centers across Europe.
The most recent European guidelines highlight that ablation should be considered as first-line therapy in selected patients with symptomatic PAF. Cryoballoon ablation is not approved as a first-line treatment in the United States.
Approximately 7 million people worldwide currently suffer from AF, an irregular quivering or rapid heart rhythm in the upper chambers of the heart that is associated with increased morbidity and mortality as well as added healthcare costs. Cryoablation involves a minimally invasive procedure that creates lesions by freezing tissue in the heart`s upper chambers, traditionally around the pulmonary veins, to block the electrical signals that trigger erratic heart rhythms. Pulmonary vein isolation (PVI) is a standard treatment for patients in the early stages of AF when the triggers come largely from these veins. However, as the disease progresses, it becomes more complicated to treat, with lower long-term success rates.
“For select patients with symptomatic paroxysmal AF and minimal or no heart disease, this therapy—with its established safety profile—has potential as a first-line treatment, providing valuable clinical insights and evidence for future therapeutic solutions,” said Heinz-Friedrich Pitschner, M.D. of the Kerckhoff-Klinik Heart Center in Germany and chair of the Cryo-FIRST steering committee.
“With the prevalence of AF expected to increase exponentially over the next generation, we believe that cryoballoon ablation therapy may safely and effectively address the disease before it progresses, improving outcomes and quality of life for patients, and reducing healthcare costs,” said Reggie Groves, vice president and general manager of Medtronic’s AF Solutions business.