01.08.15
Berkeley, Calif.-based ExThera Medical Corporation has joined a team lead by Battelle Memorial Institute that is developing a dialysis-like therapeutics device (DLT) to treat sepsis. Battelle, a nonprofit research and development organization, assembled the team in 2013 for a Defense Advanced Research Projects Agency (DARPA) contract to develop such a device for the benefit of soldiers in the battlefield. The team also includes NxStage Medical Inc., which makes products for end-stage renal failure, and Aethlon Medical Inc., which focuses on cancer, infectious disease, and other life-threatening diseases. ExThera’s specialty is bloodstream infections.
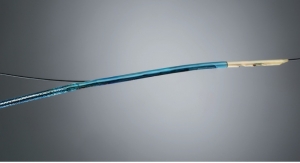
As part of the DLT Program, Battelle will perform safety and efficacy studies on ExThera’s second-generation Seraph Microbind Affinity blood filter.
“Each year there are 1.8 million new cases of bloodstream infection in the United States and Europe, causing patients serious complications and imposing a major cost burden on health care systems,” said Bob Ward, CEO of ExThera Medica. “With fewer anti-infective drugs being developed, and the continuing emergence of new drug-resistant pathogens, ExThera’s sorbent hemoperfusion device should become an important new tool for critical care clinicians.”
Initiated in 2010, DARPA’s DLT program is developing a portable system that removes “dirty” blood from the body, removes harmful agents, and returns “clean” blood to the body in a manner similar to the treatment of kidney failure with dialysis. The DLT program seeks to develop filter technologies, integrate them into a portable system, and rigorously validate filter effectiveness. The resulting multi-component device could decrease the morbidity and mortality of bloodstream infections, e.g. by preventing sepsis-induced organ failure, thereby saving thousands of lives and billions of dollars in the United States annually.
“ExThera’s novel Seraph technology, as well as cutting-edge, blood-separation technologies from other DLT developers, are needed to meet DARPA’s aggressive program requirements,” said Marty Toomajian, president, Battelle Energy, Health & Environment. “Sepsis has claimed the lives of thousands of our nation’s warfighters and the DLT system could be a major step forward in improving survival rates.”
DARPA’s integration and validation of a DLT system focuses on submitting an investigational device exemption to the FDA before the completion of the program. From there, the DLT system would be available for transition to clinical trials required for final regulatory approval. NxStage would manufacture and commercialize the DLT system while ExThera and other DLT developers would manufacture and supply blood separation filters for use in the system.
Since 2008, ExThera (short for Extracorporeal Therapy) has been pursuing a nearly identical goal within the civilian market. The Seraph, which is now fully developed and about to enter clinical trials in Europe, is a broad-spectrum hemoperfusion device (which filters blood outside the body) designed to remove a long list of pathogens and toxins from whole blood. These include viruses, drug-resistant bacteria, fungi and inflammatory cytokines. While adding nothing to the blood, Seraph uses a biomimetic adsorption media that include chemically-bonded heparin and supplemental adsorbent materials in a single cartridge. The well-known anti-thrombogenic properties of heparin are meant to contribute to the safety of the device, especially in situations when the device must be used without systemic blood thinners.
As part of the DLT Program, Battelle will perform safety and efficacy studies on ExThera’s second-generation Seraph Microbind Affinity blood filter.
“Each year there are 1.8 million new cases of bloodstream infection in the United States and Europe, causing patients serious complications and imposing a major cost burden on health care systems,” said Bob Ward, CEO of ExThera Medica. “With fewer anti-infective drugs being developed, and the continuing emergence of new drug-resistant pathogens, ExThera’s sorbent hemoperfusion device should become an important new tool for critical care clinicians.”
Initiated in 2010, DARPA’s DLT program is developing a portable system that removes “dirty” blood from the body, removes harmful agents, and returns “clean” blood to the body in a manner similar to the treatment of kidney failure with dialysis. The DLT program seeks to develop filter technologies, integrate them into a portable system, and rigorously validate filter effectiveness. The resulting multi-component device could decrease the morbidity and mortality of bloodstream infections, e.g. by preventing sepsis-induced organ failure, thereby saving thousands of lives and billions of dollars in the United States annually.
“ExThera’s novel Seraph technology, as well as cutting-edge, blood-separation technologies from other DLT developers, are needed to meet DARPA’s aggressive program requirements,” said Marty Toomajian, president, Battelle Energy, Health & Environment. “Sepsis has claimed the lives of thousands of our nation’s warfighters and the DLT system could be a major step forward in improving survival rates.”
DARPA’s integration and validation of a DLT system focuses on submitting an investigational device exemption to the FDA before the completion of the program. From there, the DLT system would be available for transition to clinical trials required for final regulatory approval. NxStage would manufacture and commercialize the DLT system while ExThera and other DLT developers would manufacture and supply blood separation filters for use in the system.
Since 2008, ExThera (short for Extracorporeal Therapy) has been pursuing a nearly identical goal within the civilian market. The Seraph, which is now fully developed and about to enter clinical trials in Europe, is a broad-spectrum hemoperfusion device (which filters blood outside the body) designed to remove a long list of pathogens and toxins from whole blood. These include viruses, drug-resistant bacteria, fungi and inflammatory cytokines. While adding nothing to the blood, Seraph uses a biomimetic adsorption media that include chemically-bonded heparin and supplemental adsorbent materials in a single cartridge. The well-known anti-thrombogenic properties of heparin are meant to contribute to the safety of the device, especially in situations when the device must be used without systemic blood thinners.