“We believe that in the vast majority of women, the procedure should not be performed,” said William Maisel, deputy director for science and chief scientist at the FDA’s Center for Devices and Radiological Health. The FDA's primary concern is the safety and well-being of patients, and taking these steps will help the agency's safety recommendations to be implemented as quickly as possible. The FDA strongly encourages doctors to inform their patients of the risk of spreading unsuspected cancer from the use of these devices in fibroid surgery and discuss the benefits and risks associated with all treatment options."
The recommended labeling revisions include two contraindications: one stating that power morcellators are contraindicated in gynecologic surgery in women with known or suspected malignancy and a similar statement pertaining to use of power morcellators to treat fibroids in peri- or postmenopausal women or those who are candidates for surgical resection.
The "black box" warning states that "uterine tissue may contain unsuspected cancer. The use of laparoscopic power morcellators during fibroid surgery may spread cancer and decrease the long-term survival of patients. This information should be shared with patients when considering surgery with the use of these devices."
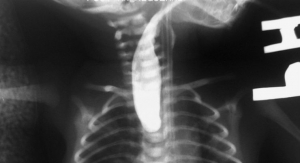
The move strengthens guidance the FDA issued in April and draws tight boundaries around use of a device that divided gynecologists and alarmed women. Morcellators were being used in thousands of minimally-invasive procedures every year to remove growths known as fibroids. While fibroids are benign, they can be hard to distinguish from a dangerous form of cancer called uterine sarcoma, which can’t be reliably detected before surgery. Morcellators, which typically use a fast-spinning blade to cut up tissue so it can be taken out through small incisions, can spread the malignancy and worsen the outcome, the FDA said.
In July an FDA advisory committee spent two days examining the risks and benefits of using power morcellators during fibroid removal but it made no specific recommendations. Shortly after the advisory committee met, a study to quantify the cancer risk associated with use of power morcellators to treat fibroids suggested one of every 368 morcellator procedures leads to dispersal of unrecognized cancer. At the July advisory committee meeting, the FDA staff report cited estimates of one in 352 for unsuspected uterine sarcoma and one in 498 for leiomyosarcoma.
The tool was first approved for gynecology in the 1990s but its popularity rose in recent years with the rise of minimally invasive and robotic surgery. The FDA began looking into the issue after a Boston, Mass., doctor, Amy Reed, went public in a December 2013 Wall Street Journal article that detailed her worsened cancer after a hysterectomy using the device.
The FDA said it wanted to leave a window open for the small number of women for whom the tool’s benefit may outweigh its risks. This includes younger women whose fertility could be preserved by removing only the fibroids and not the uterus. Still, the agency said any woman who does undergo the procedure should be warned that morcellation could spread unsuspected cancer.
The FDA still is considering other steps to reduce the risk of spreading hidden cancers from morcellation, including possible containment systems and improved preoperative detection of cancer. Some doctors have been working to develop surgical bags to surround the morcellation procedure and guard against cancer dissemination. While bags are a reasonable practice for skilled doctors, they can also introduce other risks, experts noted.