11.12.14
Ativa Medical Corp. has a new leader.
The developer of the world’s first multi-detection micro lab has named James M. McNally, Ph.D., as president and CEO.
McNally brings to Ativa more than 25 years of executive leadership experience in the medical device and diagnostic industry, including most recently serving as vice president of Quest Diagnostics’ products division, including its point-of-care companies. He has been awarded 12 patents for his healthcare and diagnostic innovations.
“In my career, I’ve had the opportunity to review hundreds of diagnostic instruments and businesses; we believe Ativa’s system will be able to deliver central lab quality results in about five minutes during a single office visit. Clinicians will be able to better diagnose and treat patients, while reducing traditional lab test costs,” McNally said. “I look forward to taking this technology through commercialization.”
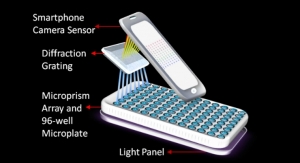
When launched, Ativa’s system should allow health care professionals to obtain results in minutes rather than hours or days, to improve the patient experience. Practitioners will get critical test information for detecting and managing disease during the clinical visit. The Ativa system’s technology, including key microfluidic components, sample preparation, disposable test cards, diagnostic tests and instrumentation, is covered by 80 U.S. patents and 184 patents worldwide.
The Ativa system also reduces the chances for lab test errors and misdiagnoses, as it does not require sample handling by multiple health care staff, the company notes. “(Central) laboratory testing is subject to many factors that potentially could adversely affect the integrity of the sample and prevent the timely reporting of an accurate test result to your provider,” according to a 2014 report by Lab Tests Online.
“Ativa’s micro lab, with the footprint of a toaster, will ultimately be able to accurately and efficiently test up to 90 percent of Medicare’s top 30 blood tests,” McNally said. “Ativa’s patented disposable microfluidic cards need only a drop of blood yet compete with the quality and consistency of large lab instruments, based on our pre-clinical complete blood count results. The Ativa system holds great potential where speed-to-diagnosis is critical, such as emergency rooms and urgent care facilities, as well as in private physician offices and retail clinics and pharmacies.”
“Jim McNally’s experience with an array of point-of-care products, including navigating commercialization issues and the FDA, gives him important insights to lead Ativa at this stage,” Board Chairman James B. Stake said.
Ativa Medical Corp. is a privately held, emerging growth medical diagnostic company developing and commercializing point-of-care diagnostic technologies. The firm is based in St. Paul, Minn.
The developer of the world’s first multi-detection micro lab has named James M. McNally, Ph.D., as president and CEO.
McNally brings to Ativa more than 25 years of executive leadership experience in the medical device and diagnostic industry, including most recently serving as vice president of Quest Diagnostics’ products division, including its point-of-care companies. He has been awarded 12 patents for his healthcare and diagnostic innovations.
“In my career, I’ve had the opportunity to review hundreds of diagnostic instruments and businesses; we believe Ativa’s system will be able to deliver central lab quality results in about five minutes during a single office visit. Clinicians will be able to better diagnose and treat patients, while reducing traditional lab test costs,” McNally said. “I look forward to taking this technology through commercialization.”
When launched, Ativa’s system should allow health care professionals to obtain results in minutes rather than hours or days, to improve the patient experience. Practitioners will get critical test information for detecting and managing disease during the clinical visit. The Ativa system’s technology, including key microfluidic components, sample preparation, disposable test cards, diagnostic tests and instrumentation, is covered by 80 U.S. patents and 184 patents worldwide.
The Ativa system also reduces the chances for lab test errors and misdiagnoses, as it does not require sample handling by multiple health care staff, the company notes. “(Central) laboratory testing is subject to many factors that potentially could adversely affect the integrity of the sample and prevent the timely reporting of an accurate test result to your provider,” according to a 2014 report by Lab Tests Online.
“Ativa’s micro lab, with the footprint of a toaster, will ultimately be able to accurately and efficiently test up to 90 percent of Medicare’s top 30 blood tests,” McNally said. “Ativa’s patented disposable microfluidic cards need only a drop of blood yet compete with the quality and consistency of large lab instruments, based on our pre-clinical complete blood count results. The Ativa system holds great potential where speed-to-diagnosis is critical, such as emergency rooms and urgent care facilities, as well as in private physician offices and retail clinics and pharmacies.”
“Jim McNally’s experience with an array of point-of-care products, including navigating commercialization issues and the FDA, gives him important insights to lead Ativa at this stage,” Board Chairman James B. Stake said.
Ativa Medical Corp. is a privately held, emerging growth medical diagnostic company developing and commercializing point-of-care diagnostic technologies. The firm is based in St. Paul, Minn.