11.07.14
Biotronik SE & Co. KG has begun enrolling the first Pulsar-18 self-expanding stent implantation patients in the investigator-initiated Bioflex Peace study. Building on positive results for the 4F stent in the earlier 4EVER and Peace trials, this new all-comers registry is designed to evaluate the Pulsar-18's efficacy for interventions on femoropopliteal lesions in a larger group of patients in a real-world setting.
The Bioflex Peace all-comers registry is a prospective, single-arm, multicenter study, which will investigate the treatment of femoropopliteal atherosclerotic lesions, following patients out to 24 months. Primary endpoints are major adverse events at six months and primary patency at 12 months. Secondary endpoints include clinical success (defined as improvement by one or more Rutherford class), target lesion revascularization (TLR), amputation rate, along with improvement in pain rating score and walking impairment. Extensive subgroup analyses are planned, including lesion location, lesion preparation prior to stenting, and stent oversizing.
"Following up on the Peace I study, which reported a primary patency rate of 79.5 percent and a freedom from TLR rate of 81 percent, Bioflex Peace will enroll up to 500 patients to evaluate the safety and efficacy of Pulsar stents in a real-world setting," said lead investigator Michael Lichtenberg, M.D., of the Vascular Center Clinic in Arnsberg, Germany. "This extensive cohort will allow subgroup analyses of different treatment strategies-including lesion preparation, the importance of correct stent sizing and spot stenting, amongst others -- with the goal of assessing the efficacy of these respective approaches."
"We are very keen to see how the Pulsar stent performs in the variously confined subgroups," said Alexander Uhl, Ph.D., Biotronik's vice president of marketing, Vascular Intervention. "Due to its design, the Pulsar stent has so far consistently demonstrated favorable patency as compared to thick strut stents, but with the additional advantage of lower profile, lower wound complication rates, reduced procedure cost and faster ambulation time. We look forward to the data regarding our Pulsar stent's performance in a much wider cohort of patients, which should further confirm the benefits of a 'low-profile' approach for lower limb interventions."
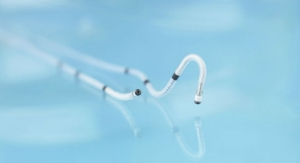
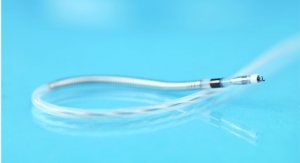
Pulsar stents feature a highly flexible design coated with proBIO, a silicon carbide layer that improves the stent's hemocompatibility and biocompatibility, which is believed to contribute to its excellent clinical results. Pulsar-18 is available in diameters of 4 mm to 7 mm and lengths of 20 mm to 200 mm, all deliverable through a 4F sheath.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.
The Bioflex Peace all-comers registry is a prospective, single-arm, multicenter study, which will investigate the treatment of femoropopliteal atherosclerotic lesions, following patients out to 24 months. Primary endpoints are major adverse events at six months and primary patency at 12 months. Secondary endpoints include clinical success (defined as improvement by one or more Rutherford class), target lesion revascularization (TLR), amputation rate, along with improvement in pain rating score and walking impairment. Extensive subgroup analyses are planned, including lesion location, lesion preparation prior to stenting, and stent oversizing.
"Following up on the Peace I study, which reported a primary patency rate of 79.5 percent and a freedom from TLR rate of 81 percent, Bioflex Peace will enroll up to 500 patients to evaluate the safety and efficacy of Pulsar stents in a real-world setting," said lead investigator Michael Lichtenberg, M.D., of the Vascular Center Clinic in Arnsberg, Germany. "This extensive cohort will allow subgroup analyses of different treatment strategies-including lesion preparation, the importance of correct stent sizing and spot stenting, amongst others -- with the goal of assessing the efficacy of these respective approaches."
"We are very keen to see how the Pulsar stent performs in the variously confined subgroups," said Alexander Uhl, Ph.D., Biotronik's vice president of marketing, Vascular Intervention. "Due to its design, the Pulsar stent has so far consistently demonstrated favorable patency as compared to thick strut stents, but with the additional advantage of lower profile, lower wound complication rates, reduced procedure cost and faster ambulation time. We look forward to the data regarding our Pulsar stent's performance in a much wider cohort of patients, which should further confirm the benefits of a 'low-profile' approach for lower limb interventions."
Pulsar stents feature a highly flexible design coated with proBIO, a silicon carbide layer that improves the stent's hemocompatibility and biocompatibility, which is believed to contribute to its excellent clinical results. Pulsar-18 is available in diameters of 4 mm to 7 mm and lengths of 20 mm to 200 mm, all deliverable through a 4F sheath.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.