10.09.14
The U.S. Food and Drug Administration (FDA) Circulatory System Devices Panel of the Medical Devices Advisory Committee has voted in favor of Boston Scientific’s Watchman left atrial appendage closure (LAAC) device.
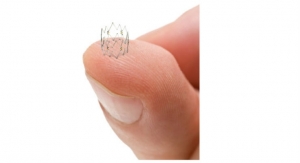
The device is a catheter-delivered heart implant designed to close the left atrial appendage in order to prevent the migration of blood clots from the there, thus reducing the incidence of stroke and systemic embolism for higher risk patients with non-valvular atrial fibrillation (AF). The LAA is a thin, sack-like appendix arising from the heart and is believed to be the source of a majority of stroke-causing blood clots in people with AF. The Watchman device was developed by Atritech, which Boston Scientific acquired in March 2011.
The panel of 12 voted unanimously that the device is safe. On the question of reasonable assurance of effectiveness, the panel vote was unfavorably (6 yes to 7 no). It provided substantial input and guidance related to the proposed indications for use and target patient population. There was general agreement among the panel members that the device provides a much-needed alternative to long-term anticoagulation for some patients.
While not bound by this vote, the FDA takes advisory panel comments and recommendations into account when reviewing the device approval application.
“There is a strong clinical need for a proven device alternative to long-term warfarin therapy for my high stroke risk patients with non-valvular atrial fibrillation,” said Vivek Reddy, M.D., director of the cardiac arrhythmia service at Mount Sinai Medical Center and co-principal investigator of the Protect AF and Prevail studies. “I’m encouraged that the panel recognized the importance of having the Watchman device as an option for appropriate patients.”
The committee’s positive vote followed a review of the most recent clinical data and analysis from two randomized control trials, Protect AF and Prevail, as well as from the CAP (continued access protocol) and CAP2 registries. Boston Scientific’s device has long-term clinical data from over 2,400 patients and nearly 6,000 patient-years of follow-up in clinical studies. It was approved for sale in Europe in 2005 and is currently approved in more than 70 countries; it is currently an investigational device in the United States.
“Today’s recommendation by the panel is another step toward making this innovative technology available to high risk patients with non-valvular atrial fibrillation who are eligible for warfarin, but who have reasons to seek an alternative to long-term therapy,” said Kenneth Stein, M.D., chief medical officer of rhythm management for Boston Scientific. “We continue to believe that the totality of the data for the Watchman Device provide reasonable assurance of its safety and efficacy as a treatment alternative for these patients. We look forward to our ongoing discussions with FDA.”
Marlborough, Mass.-based Boston Scientific Corporation makes devices for use in interventional medicine.
The device is a catheter-delivered heart implant designed to close the left atrial appendage in order to prevent the migration of blood clots from the there, thus reducing the incidence of stroke and systemic embolism for higher risk patients with non-valvular atrial fibrillation (AF). The LAA is a thin, sack-like appendix arising from the heart and is believed to be the source of a majority of stroke-causing blood clots in people with AF. The Watchman device was developed by Atritech, which Boston Scientific acquired in March 2011.
The panel of 12 voted unanimously that the device is safe. On the question of reasonable assurance of effectiveness, the panel vote was unfavorably (6 yes to 7 no). It provided substantial input and guidance related to the proposed indications for use and target patient population. There was general agreement among the panel members that the device provides a much-needed alternative to long-term anticoagulation for some patients.
While not bound by this vote, the FDA takes advisory panel comments and recommendations into account when reviewing the device approval application.
“There is a strong clinical need for a proven device alternative to long-term warfarin therapy for my high stroke risk patients with non-valvular atrial fibrillation,” said Vivek Reddy, M.D., director of the cardiac arrhythmia service at Mount Sinai Medical Center and co-principal investigator of the Protect AF and Prevail studies. “I’m encouraged that the panel recognized the importance of having the Watchman device as an option for appropriate patients.”
The committee’s positive vote followed a review of the most recent clinical data and analysis from two randomized control trials, Protect AF and Prevail, as well as from the CAP (continued access protocol) and CAP2 registries. Boston Scientific’s device has long-term clinical data from over 2,400 patients and nearly 6,000 patient-years of follow-up in clinical studies. It was approved for sale in Europe in 2005 and is currently approved in more than 70 countries; it is currently an investigational device in the United States.
“Today’s recommendation by the panel is another step toward making this innovative technology available to high risk patients with non-valvular atrial fibrillation who are eligible for warfarin, but who have reasons to seek an alternative to long-term therapy,” said Kenneth Stein, M.D., chief medical officer of rhythm management for Boston Scientific. “We continue to believe that the totality of the data for the Watchman Device provide reasonable assurance of its safety and efficacy as a treatment alternative for these patients. We look forward to our ongoing discussions with FDA.”
Marlborough, Mass.-based Boston Scientific Corporation makes devices for use in interventional medicine.