09.02.14
In a boon to stem cell research and regenerative medicine, scientists at Boston Children’s Hospital, the Wyss Institute for Biologically Inspired Engineering at Harvard University and Boston University have created a computer algorithm called CellNet as a “roadmap” for cell and tissue engineering, to ensure that cells engineered in the lab have the same favorable properties as cells in our own bodies. CellNet and its application to stem cell engineering are described in two back-to-back papers in the August 14 issue of the journal Cell.
Globally, scientists are engaged in culturing pluripotent stem cells (capable of forming all the body’s tissues) and transforming them into specialized cell types for use in research and regenerative medicine. Available as an Internet resource for any scientist to use, CellNet provides a much needed “quality assurance” measure for this work.
The two papers also clarify uncertainty around which methods are best for stem cell engineering, and should advance the use of cells derived from patient tissues to model disease, test potential drugs and use as treatments. For example, using CellNet, one of the studies unexpectedly found that skin cells can be converted into intestinal cells that were able to reverse colitis in a mouse model.
“To date, there has been no systematic means of assessing the fidelity of cellular engineering—to determine how closely cells made in a petri dish approximate natural tissues in the body,” said George Q. Daley, M.D., Ph.D., director of the stem cell transplantation program at Boston Children’s and senior investigator on both studies. “CellNet was developed to assess the quality of engineered cells and to identify ways to improve their performance.”
Gene Signatures
CellNet applies network biology to discover the complex network of genes that are turned on or off in an engineered cell, known as the cell’s Gene Regulatory Network or GRN. It then compares that network to the cell’s real-life counterpart in the body, as determined from public genome databases. Through this comparison, researchers can rigorously and reliably assess:
“CellNet will also be a powerful tool to advance synthetic biology—to engineer cells for specific medical applications,” says James Collins, Ph.D., core faculty member at the Wyss Institute and the William F. Warren distinguished professor at Boston University, co-senior investigator on one of the studies.
The CellNet platform “can be applied to almost any study and allows users to refine the engineering process,” said an announcement from Mayo Clinic, where one of the first coauthors Hu Li, Ph.D. is based. “In the long run, it should provide a reliable shortcut to the early phases of drug development, individualized cancer therapies and pharmacogenomics.”
“Based on the results, you can analyze why your cell types are not similar to the expected target cells,” Li said. That will allow research teams to more quickly refine their methods and develop the type of cell lines they must acquire prior to further research.
Mayo Clinic will provide open access CellNet.
Globally, scientists are engaged in culturing pluripotent stem cells (capable of forming all the body’s tissues) and transforming them into specialized cell types for use in research and regenerative medicine. Available as an Internet resource for any scientist to use, CellNet provides a much needed “quality assurance” measure for this work.
The two papers also clarify uncertainty around which methods are best for stem cell engineering, and should advance the use of cells derived from patient tissues to model disease, test potential drugs and use as treatments. For example, using CellNet, one of the studies unexpectedly found that skin cells can be converted into intestinal cells that were able to reverse colitis in a mouse model.
“To date, there has been no systematic means of assessing the fidelity of cellular engineering—to determine how closely cells made in a petri dish approximate natural tissues in the body,” said George Q. Daley, M.D., Ph.D., director of the stem cell transplantation program at Boston Children’s and senior investigator on both studies. “CellNet was developed to assess the quality of engineered cells and to identify ways to improve their performance.”
Gene Signatures
CellNet applies network biology to discover the complex network of genes that are turned on or off in an engineered cell, known as the cell’s Gene Regulatory Network or GRN. It then compares that network to the cell’s real-life counterpart in the body, as determined from public genome databases. Through this comparison, researchers can rigorously and reliably assess:
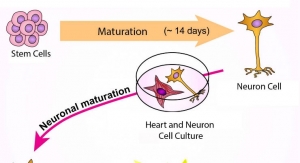
- The quality of induced pluripotent stem cells (iPS cells) made by reprogramming blood cells or skin cells;
- The quality of specialized cells—such as liver, heart, muscle, brain or blood cells—made from either iPS cells or embryonic stem cells;
- The quality of specialized cells made from other specialized cells (such as liver cells made directly from skin cells); and
- What specific improvements need to be made to the engineering process.
“CellNet will also be a powerful tool to advance synthetic biology—to engineer cells for specific medical applications,” says James Collins, Ph.D., core faculty member at the Wyss Institute and the William F. Warren distinguished professor at Boston University, co-senior investigator on one of the studies.
The CellNet platform “can be applied to almost any study and allows users to refine the engineering process,” said an announcement from Mayo Clinic, where one of the first coauthors Hu Li, Ph.D. is based. “In the long run, it should provide a reliable shortcut to the early phases of drug development, individualized cancer therapies and pharmacogenomics.”
“Based on the results, you can analyze why your cell types are not similar to the expected target cells,” Li said. That will allow research teams to more quickly refine their methods and develop the type of cell lines they must acquire prior to further research.
Mayo Clinic will provide open access CellNet.