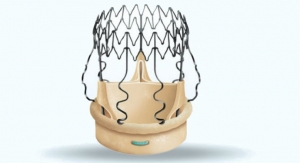
Santa Rosa, Calif.-based Direct Flow Medical Inc., a transcatheter heart valve developer, has received CE mark for a 23-millimeter (mm) sized valve as part of its Transcatheter Aortic Valve System. The additional CEM mark expands the patient population that can be treated with its technology. The company also announced receipt of the CE mark for implantation of all of its valves without the use of contrast media, protecting patients from kidney injury during transcatheter aortic valve implantation (TAVI).
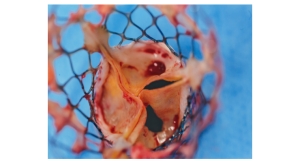
The company now has a valve portfolio that includes 23 mm, 25 mm, 27 mm and 29 mm valves, which can treat patients with annulus sizes from 19 mm to 28 mm. The company's valves feature a double-ring design that conforms to the anatomy and creates a tight and durable seal around the annulus. According to the company, the valves allow for "complete assessment of hemodynamic performance and unlimited repositioning until optimal results are obtained."
All valves can be delivered through the same delivery system.
“These approvals are a testament to our continuous focus on innovation and patient outcomes. Physicians will not only be able to treat a broader patient population, but will also be able to implant this technology with no contrast and no compromises,” said Bernard Lyons, CEO of Direct Flow Medical. “Our pipeline of structural heart technologies is strong and we continue to invest in new products and new indications for our unique therapeutic platform.”
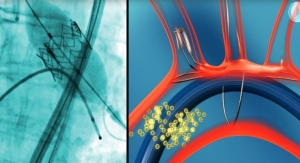
According to company officials, the valve is designed to improve TAVI outcomes by eliminating aortic regurgitation (blood flow back into the valve), offering the ability to reposition during the procedure and reducing overall complications.
Acute kidney injury occurs in about 22 percent of patients undergoing TAVI, increasing their risk of peri-procedural complications, prolonged hospitalization and mortality.
“Use of contrast during angiographic procedures is a well-documented cause of acute kidney injury,” said Direct Flow Medical Chief Medical Officer Charles Davidson, M.D. “By eliminating or reducing the quantity of contrast administered during TAVI, we will be able to better protect the kidneys and minimize peri-procedural complications.”
The Direct Flow Medical Transcatheter Aortic Valve System is commercially available in Europe. In the United States, the company is pursuing regulatory approval and currently is enrolling patients in its SALUS pivotal trial.
Founded in 2004, Direct Flow Medical's manufacturing facilities are in Lake Forest, Calif. The Company’s proprietary technology is not limited to aortic valve disease, and is applicable to mitral and other heart valve anatomical sites, the company claims. The company is venture-backed.