07.18.14
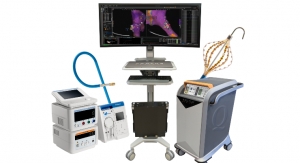
Covidien plc has released its new Emprint ablation system with Thermosphere technology, an advanced ablation system designed to precisely heat and destroy diseased soft tissue in the liver, lungs and kidney.
The Emprint ablation system is designed to deliver precise ablation directly to soft tissue, including liver tumors. This technology is hoped to ensure more certainty regardless of the target location or tissue type, thus making liver tumor ablation more accurate.
Covidien’s ablation system provides clinicians with three kinds of spatial energy control—thermal, field and wavelength. This helps them to create predictable and spherical ablation zones regardless of target location, tissue type or changes in tissue properties during a procedure.
By providing predictable spherical ablation zones, this technology offers physicians more choices in terms of approach, further simplifying needle placement and saving planning and procedure time.
A liver tumor ablation procedure destroys the tumor without removing it, thus providing an alternative to open surgery resection or other treatments. This method is ideal for patients for whom surgical removal of tumors is not an optimal solution.
With Covidien’s ablation system, physicians can treat liver tumor by non-surgical procedures. They utilize ultrasound or CT (computed tomography) scanning to guide the system’s antenna and insert it directly into the tumor through the skin. Apart from non-surgical procedures, Covidien’s advanced ablation system can also be used for minimally invasive surgery and open surgery.
Covidien received the U.S. Food and Drug Administration 510(k) clearance for Emprint in April this year. The company has also completed all European requirements for attaining the CE mark for the product. Covidien expects to fully launch the Emprint Ablation System in the United States and the European Union in the present quarter.
The Emprint ablation system is designed to deliver precise ablation directly to soft tissue, including liver tumors. This technology is hoped to ensure more certainty regardless of the target location or tissue type, thus making liver tumor ablation more accurate.
Covidien’s ablation system provides clinicians with three kinds of spatial energy control—thermal, field and wavelength. This helps them to create predictable and spherical ablation zones regardless of target location, tissue type or changes in tissue properties during a procedure.
By providing predictable spherical ablation zones, this technology offers physicians more choices in terms of approach, further simplifying needle placement and saving planning and procedure time.
A liver tumor ablation procedure destroys the tumor without removing it, thus providing an alternative to open surgery resection or other treatments. This method is ideal for patients for whom surgical removal of tumors is not an optimal solution.
With Covidien’s ablation system, physicians can treat liver tumor by non-surgical procedures. They utilize ultrasound or CT (computed tomography) scanning to guide the system’s antenna and insert it directly into the tumor through the skin. Apart from non-surgical procedures, Covidien’s advanced ablation system can also be used for minimally invasive surgery and open surgery.
Covidien received the U.S. Food and Drug Administration 510(k) clearance for Emprint in April this year. The company has also completed all European requirements for attaining the CE mark for the product. Covidien expects to fully launch the Emprint Ablation System in the United States and the European Union in the present quarter.