07.15.14
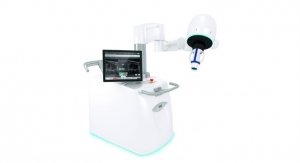
Theraclion, a French company specializing in advanced medical equipment for echotherapy, has been granted investigational device exemption from the U.S. Food and Drug Administration (FDA) to evaluate the treatment of breast fibroadenoma with its Echopulse device.
In collaboration with the University of Virginia, the study will be conducted on 20 patients with breast fibroadenoma and is designed to collect data on the safety and efficacy of the Echopulse, a device that utilizes high-intensity focused ultrasound (HIFU) treatment guided by ultrasound imaging. The trial is the first phase of the product marketing application in the United states and will be followed by a multicenter study.
Benign breast tumors constitute a growing percentage of detected breast pathologies, with an estimated 10 percent of women developing a fibroadenoma in their lifetime. A market survey of the diagnosis and therapy of breast tumors (performed by Life Science Intelligence in 2007) showed that more than 1.3 million cases of breast fibroadenoma were diagnosed in 2006, and more than 50 percent of those cases needed therapeutic excision. The same survey estimated that 1.5 million cases of breast fibroadenoma would be diagnosed in 2012.
The Echopulse treats breast fibroadenomas without significant side effects for the patient, with scar-free, non-invasive removal. The absence of skin damage reduces the likelihood of post-operative infection. The outpatient procedure is performed under local anesthesia or conscious sedation, enabling patients to immediately resume their normal activities. The flexibility of the process and the absence of hospitalization can reduce costs and optimize care management, the company claims.
The Echopulse technology has received the CE Mark in the European Union for the treatment of breast fibroadenoma and benign thyroid nodules. Therclion hopes to market the device in the Middle East, Africa, Asia and Latin America as well. Registration in China and the United States are expected by the end of 2015 and 2017, respectively.
"We are delighted with this approval from the FDA, which marks the first step in the clinical trial and marketing application process for Echopulse in the United States," CEO Stefano Vagliani said.
Echotherapy is a new non-invasive therapeutic approach that involves the use of HIFU to produce very localized ablation without skin damage. The ultrasound is focused on a previously defined zone within which the increase in temperature causes necrosis of the targeted tissues. This avoids damage to the healthy tissue around the lesion. Echotherapy enables tumors (such as thyroid nodules and breast fibroadenoma) to be simultaneously viewed and treated without incisions or scarring.
Based in Paris, France, Theraclion develops non-invasive HIFU treatments for benign tumors. The company's main investor is Truffle Capital and also receives funding from Bpifrance (formerly OSEO, the French state innovation agency).
In collaboration with the University of Virginia, the study will be conducted on 20 patients with breast fibroadenoma and is designed to collect data on the safety and efficacy of the Echopulse, a device that utilizes high-intensity focused ultrasound (HIFU) treatment guided by ultrasound imaging. The trial is the first phase of the product marketing application in the United states and will be followed by a multicenter study.
Benign breast tumors constitute a growing percentage of detected breast pathologies, with an estimated 10 percent of women developing a fibroadenoma in their lifetime. A market survey of the diagnosis and therapy of breast tumors (performed by Life Science Intelligence in 2007) showed that more than 1.3 million cases of breast fibroadenoma were diagnosed in 2006, and more than 50 percent of those cases needed therapeutic excision. The same survey estimated that 1.5 million cases of breast fibroadenoma would be diagnosed in 2012.
The Echopulse treats breast fibroadenomas without significant side effects for the patient, with scar-free, non-invasive removal. The absence of skin damage reduces the likelihood of post-operative infection. The outpatient procedure is performed under local anesthesia or conscious sedation, enabling patients to immediately resume their normal activities. The flexibility of the process and the absence of hospitalization can reduce costs and optimize care management, the company claims.
The Echopulse technology has received the CE Mark in the European Union for the treatment of breast fibroadenoma and benign thyroid nodules. Therclion hopes to market the device in the Middle East, Africa, Asia and Latin America as well. Registration in China and the United States are expected by the end of 2015 and 2017, respectively.
"We are delighted with this approval from the FDA, which marks the first step in the clinical trial and marketing application process for Echopulse in the United States," CEO Stefano Vagliani said.
Echotherapy is a new non-invasive therapeutic approach that involves the use of HIFU to produce very localized ablation without skin damage. The ultrasound is focused on a previously defined zone within which the increase in temperature causes necrosis of the targeted tissues. This avoids damage to the healthy tissue around the lesion. Echotherapy enables tumors (such as thyroid nodules and breast fibroadenoma) to be simultaneously viewed and treated without incisions or scarring.
Based in Paris, France, Theraclion develops non-invasive HIFU treatments for benign tumors. The company's main investor is Truffle Capital and also receives funding from Bpifrance (formerly OSEO, the French state innovation agency).