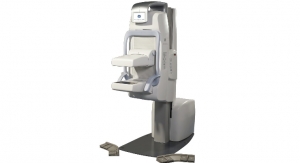
According to the results of a groundbreaking study published in the June 25 issue of the Journal of the American Medical Association (JAMA), 3-D mammography (breast tomosynthesis) screening technology made by Bedford, Mass.-based Hologic Inc. finds significantly more invasive cancers than a traditional mammogram. The researchers also found that 3-D mammography reduces the number of women called back for unnecessary testing due to false alarms, which--according to the study's authors—reduces anxiety, as well as healthcare costs.
The study, "Breast Cancer Screening Using Tomosynthesis in Combination with Digital Mammography," was led by Sarah M. Friedewald, M.D., of the Caldwell Breast Center, Advocate Lutheran General Hospital in Park Ridge, Ill. A total of 454,850 examinations (281,187 conventional mammograms compared to 173,663 3-D mammography exams) were included in the study. The study was the largest to date, involving 139 doctors from 13 U.S. academic and community-based sites
Significant findings include:
• A 41 percent increase in the detection of invasive breast cancers;
• A 29 percent increase in the detection of all breast cancers;
• A 15 percent decrease in women recalled for additional imaging;
• A 49 percent increase in positive predictive value (PPV) for a recall. (PPV for recall is a widely used measure of the proportion of women recalled from screening that are found to have breast cancer. The PPV for a recall increased from 4.3 to 6.4 percent);
• A 21 percent increase in PPV for biopsy. (PPV for biopsy is a widely used measure of the proportion of women having a breast biopsy that are found to have breast cancer. The PPV for a breast biopsy increased from 24.2 to 29.2 percent); and
• No significant change in the detection of ductal carcinoma in situ (DCIS). (DCIS is a non-invasive cancer. It has not spread beyond the milk duct into any normal surrounding breast tissue.)
"The JAMA 3-D study validates the findings of previously published studies but on a much larger scale," said Peter J. Valenti III , Hologic division president, Breast and Skeletal Health Solutions. "The study addresses the two most frequently cited concerns with breast cancer screening—that we are finding too many cancers that don't need to be treated and that too many women are being called back for unnecessary additional testing. Each of the outcomes measured was statistically significant and reinforced the benefits of Hologic 3-D mammography in addressing these challenges."
Five leading academic hospitals participated in the study: Massachusetts General Hospital; Yale University School of Medicine in Connecticut ; University Hospitals Case Medical Center in Ohio; Albert Einstein Healthcare Network; and the Perelman School of Medicine of the University of Pennsylvania in Pennsylvania.
Eight community-based sites participated in the study: Caldwell Breast Center of Advocate Lutheran General Hospital in Illinois; TOPS Comprehensive Breast Center in Texas; Washington Radiology Associates, P.C. in Washington, D.C. ; Radiology Associates of Hollywood and Memorial Healthcare System in Florida; Evergreen Health Breast Center and Radia Inc. P.S. in the state of Washington; Edith Sanford Breast Health Institute in South Dakota; Invision Sally Jobe Breast Centers and Radiology Imaging Associates in Colorado; and John C. Lincoln Breast Health and Research Center in Arizona.
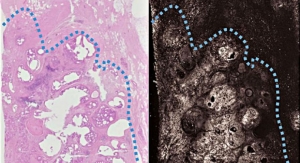
While digital (two-dimensional) mammography is considered one of the most advanced breast cancer screening technologies available today, it provides a "flat' picture of the breast. The breast is a three-dimensional object composed of different structures, such as blood vessels, milk ducts, fat, and ligaments. Such structures, which are located at different heights within the breast, can overlap and cause confusion when viewed as a two-dimensional, flat image. This confusion of overlapping tissue is a leading reason why small breast cancers may be missed and normal tissue may appear abnormal, leading to unnecessary call backs.
According to Hologic, its 3-D mammography system is the first and currently the only U.S. Food and Drug Administration-approved 3-D mammography system in the United States. The technology was approved for breast cancer screening and diagnosis in February 2011 and has been available in countries recognizing the CE mark since 2008.
An estimated 6 million women in the U.S. will be screened with the technology in 2014.
Hologic makes diagnostic products, medical imaging systems and surgical products focused on breast health, diagnostics, gynecological, surgical, and skeletal health.