06.18.14
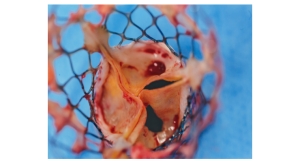
The U.S. Food and Drug Administration (FDA) has approved the CoreValve transcatheter aortic valve replacement (TAVR) from Medtronic Inc. for use in more patients.
CoreValve, though on the market for a number of years in Europe (since 2007), finally was approved in the United States in January for use in patients designated as facing extreme risk of death if they underwent open-heart surgery, the standard procedure for valve replacement. Now, doctors can use CoreValve in patients designated at high risk of death through surgery, a broader patient population.
Medtronic and Edwards Lifesciences Corp. have divvied up the TAVR market—in the United States and abroad. Companies such as St. Jude Medical Inc. and Boston Scientific Corp. and in the process of releasing their first-generation devices in the United States. Other smaller companies also are trying to get into the mix. Last month, Medtronic and Edwards settled an ongoing patent dispute regarding the valves with a cross-license deal and agreement by Medtronic to pay Edwards about $1.1 billion through 2022, with $750 million upfront. Edwards also recently received an FDA approval for its next-generation Sapien XL valve.
The FDA approved CoreValve without the need for an independent device advisory panel review due to results from a clinical trial comparing TAVR with CoreValve to traditional surgical aortic valve replacement. Survival at one year for patients receiving CoreValve was 85.8 percent, which was statistically superior to patients receiving a surgical valve at 80.9 percent.
"This rigorous trial has defined a new standard for transcatheter valve performance, with superiority results that give physicians even more confidence in making TAVR treatment decisions," said David H. Adams, M.D., chair of the Department of Cardiothoracic Surgery at the Mount Sinai Hospital, New York, N.Y., national co-principal investigator of the CoreValve U.S. pivotal trial. "With this approval, we can treat more patients due to the broad range of CoreValve sizes, and we have an option compared to surgery that provides a greater chance for a longer life while minimizing the risk of stroke."
Roughly 550,000 people worldwide receive a diagnosis of severe aortic stenosis, a condition in which calcium buildup in a valve can prevent the heart from pumping blood properly. About 100,000 of those are Americans and, of them, about 35,000 are designated at extreme or high risk of death from open-heart surgery. Half of the people who are diagnosed with the condition die within two years of the diagnosis.
"It's rewarding that we can now offer this life-saving therapy to patients at increased risk for surgery," said John Liddicoat, M.D., senior vice president, Medtronic, and president of the Medtronic Structural Heart Business. "There is a lot of excitement among U.S. heart teams for the CoreValve System's high risk approval, and its unique design that leads to the clinical outcomes seen in the high-risk trial. We will continue to safely introduce CoreValve System to these physicians, supporting heart teams through comprehensive training and education, imaging and patient evaluation programs."
CoreValve, though on the market for a number of years in Europe (since 2007), finally was approved in the United States in January for use in patients designated as facing extreme risk of death if they underwent open-heart surgery, the standard procedure for valve replacement. Now, doctors can use CoreValve in patients designated at high risk of death through surgery, a broader patient population.
Medtronic and Edwards Lifesciences Corp. have divvied up the TAVR market—in the United States and abroad. Companies such as St. Jude Medical Inc. and Boston Scientific Corp. and in the process of releasing their first-generation devices in the United States. Other smaller companies also are trying to get into the mix. Last month, Medtronic and Edwards settled an ongoing patent dispute regarding the valves with a cross-license deal and agreement by Medtronic to pay Edwards about $1.1 billion through 2022, with $750 million upfront. Edwards also recently received an FDA approval for its next-generation Sapien XL valve.
The FDA approved CoreValve without the need for an independent device advisory panel review due to results from a clinical trial comparing TAVR with CoreValve to traditional surgical aortic valve replacement. Survival at one year for patients receiving CoreValve was 85.8 percent, which was statistically superior to patients receiving a surgical valve at 80.9 percent.
"This rigorous trial has defined a new standard for transcatheter valve performance, with superiority results that give physicians even more confidence in making TAVR treatment decisions," said David H. Adams, M.D., chair of the Department of Cardiothoracic Surgery at the Mount Sinai Hospital, New York, N.Y., national co-principal investigator of the CoreValve U.S. pivotal trial. "With this approval, we can treat more patients due to the broad range of CoreValve sizes, and we have an option compared to surgery that provides a greater chance for a longer life while minimizing the risk of stroke."
Roughly 550,000 people worldwide receive a diagnosis of severe aortic stenosis, a condition in which calcium buildup in a valve can prevent the heart from pumping blood properly. About 100,000 of those are Americans and, of them, about 35,000 are designated at extreme or high risk of death from open-heart surgery. Half of the people who are diagnosed with the condition die within two years of the diagnosis.
"It's rewarding that we can now offer this life-saving therapy to patients at increased risk for surgery," said John Liddicoat, M.D., senior vice president, Medtronic, and president of the Medtronic Structural Heart Business. "There is a lot of excitement among U.S. heart teams for the CoreValve System's high risk approval, and its unique design that leads to the clinical outcomes seen in the high-risk trial. We will continue to safely introduce CoreValve System to these physicians, supporting heart teams through comprehensive training and education, imaging and patient evaluation programs."