Aortic regurgitation is a condition that occurs when your heart's aortic valve doesn't close tightly. Aortic valve regurgitation allows some of the blood that was just pumped out of your heart's main pumping chamber, the left ventricle, to leak back into it.
The system addresses this by sealing the annulus and enabling complete assessment of hemodynamic performance with unlimited repositioning of the valve after full deployment. The system avoids rapid pacing of the heart during deployment and post-dilatation following placement, minimizing the risk of hemodynamic instability for patients, according to the company.
The aortic surgical annulus is a complex three-dimensional crown shaped structure. The diameter at the base of the aortic root, the basal ring, is also called the “aortic annulus,” specifically during surgical aortic valve sizing. The basal ring is not a true anatomical entity but is defined as a virtual ring with three anchors at the nadir of each of the attachments of the aortic cusps. The basal ring is non-circular and may have an oval or elliptical shape, and calcification makes its shape non-homogenous.
During surgical aortic valve replacement, the native aortic leaflets are cut out along the line of the surgical annulus, but the sizing of the “annulus” is done under direct vision by selecting an aortic valve sizer that best fits the size of the basal ring after the native aortic leaflets are removed. The sizers are unique to the type of prostheses used and are not interchangeable. The surgically implanted prosthesis is then sewn into place and this is one of the biggest differences between a surgically implanted aortic valve and an aortic valve implanted using transcatheter techniques.
The surgically implanted prosthesis is sewn into place at the level of the surgical annulus between the basal ring and the sino-tubular junction. The transcatheter aortic valve is anchored at the basal ring calcification without any sutures.1
The primary endpoint of the trial is a composite of all-cause mortality and disabling stroke at 12 months. Principal investigators for the trial are Murat Tuzcu, M.D., vice chairman of the Department of Cardiology, Cleveland Clinic, and Patrick McCarthy, M.D., director of the Bluhm Cardiovascular Institute and chief of cardiac surgery, Northwestern Memorial Hospital.
An earlier feasibility phase of the Salus trial conducted in 2013 evaluated the system in 30 patients. The 30-day outcomes were presented today by Tuzcu at the EuroPCR conference, demonstrating a survival rate of 97 percent, low procedural complications, no incidence of stroke, a three percent rate of permanent pacing and 100 percent of patients with mild or less aortic regurgitation. The mean aortic gradient decreased from 44.5 mmHg to 12.7 mmHg at 30 days.
“We have been working closely with the FDA since the early development of the Direct Flow Medical technology and it has been a collaborative effort with the FDA which has led to an approval to start the pivotal phase of the Salus trial. The technology has shown outstanding performance in clinical trial and commercial settings, significantly reducing the risk of aortic regurgitation and improving patient survival. We look forward to expanding the clinical use to additional centers in the U.S. and continuing to obtain the same strong outcomes in the pivotal trial,” said Direct Flow Medical Chief Medical Officer Charles Davidson, M.D.
The Direct Flow Medical system received the CE mark in January 2013 and is currently available commercially in Europe.
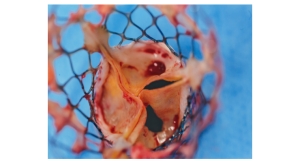
Rather than a metal stent, the Direct Flow's system incorporates a polymer frame, which is expanded using pressurized saline and contrast for placement, assessment and repositioning. The saline/contrast solution is easily exchanged for a quick-curing polymer that solidifies and secures the valve in place once optimal positioning is reached. The unique double-ring design of the valve creates a tight seal around the annulus. The system is fully repositionable and retrievable up until polymer exchange. The metal-free design enables a low-profile (18 French), fully sheathed delivery system for all valve sizes that minimizes vascular complications and improves hemodynamic outcomes.
Founded in 2004, Direct Flow Medical is headquartered in Santa Rosa, Calif., with technology and manufacturing facilities in Lake Forest, Calif.
1. George Gellert, M.D., Medical Director, Interventional Echocardiography. Structural Heart Program. The Cavanagh Heart Center. Banner Good Samaritan Medical Center