05.02.14
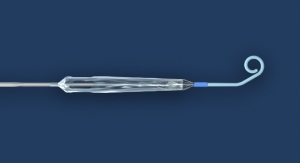
Stent developer Tryton Medical Inc. has received CE Mark for the treatment of Left Main Coronary artery disease. The company claims it is the first to earn a CE Mark for this indication.
Left main disease, an accumulation of plaque that narrows the base of the coronary tree, is a persistent challenge in interventional cardiology, as more than 75 percent of left main lesions are bifurcations. There are approximately 200,000 left main cardiac surgeries performed annually.
"A predictable and safe outcome is essential for the treatment of this high-risk population. The Tryton Side Branch stent provides the necessary control in each step of the procedure,” said Robert-Jan van Geuns, M.D., Ph.D., of Erasmus MC (Rotterdam, the Netherlands). “With the launch last summer of the Tryton Short stent, the range of Tryton stents allows me to treat the vast majority of my left main bifurcation lesions cases with a predictable procedure and durable result."
"Tryton Medical is dedicated to the treatment of all coronary bifurcations. Obtaining CE-mark approval for the left main indication significantly expands the market opportunity for our stent platform," Tryton CEO Shawn McCarthy said. "We continue to invest and introduce product innovation, clinical evidence, and physician education to advance the standard of care for bifurcated coronary artery disease."
The Tryton Side Branch Stent is commercially available in Europe and parts of the Middle East, is investigational in the United States, and is not available in Japan.
Coronary artery disease often results in the buildup of plaque at the site of a bifurcation, where one artery branches from another. Current approaches to treating these lesions are time consuming and technically difficult. As a result, the side branch is often left unstented, leaving it vulnerable to higher rates of restenosis, the re-narrowing of the stented vessel following implantation.
Tryton Medical develops stent systems for the treatment of bifurcation lesions. The company was founded in 2003 in Durham, N.C., by Aaron V. Kaplan, M.D. (professor of medicine at Dartmouth Medical School/Dartmouth-Hitchcock Medical Center) and Dan Cole, general partner at Spray Ventures. Privately held, Tryton is backed by PTV Sciences, RiverVest Venture Partners, Spray Venture Partners, and the 3x5 Special Opportunity Fund.
Left main disease, an accumulation of plaque that narrows the base of the coronary tree, is a persistent challenge in interventional cardiology, as more than 75 percent of left main lesions are bifurcations. There are approximately 200,000 left main cardiac surgeries performed annually.
"A predictable and safe outcome is essential for the treatment of this high-risk population. The Tryton Side Branch stent provides the necessary control in each step of the procedure,” said Robert-Jan van Geuns, M.D., Ph.D., of Erasmus MC (Rotterdam, the Netherlands). “With the launch last summer of the Tryton Short stent, the range of Tryton stents allows me to treat the vast majority of my left main bifurcation lesions cases with a predictable procedure and durable result."
"Tryton Medical is dedicated to the treatment of all coronary bifurcations. Obtaining CE-mark approval for the left main indication significantly expands the market opportunity for our stent platform," Tryton CEO Shawn McCarthy said. "We continue to invest and introduce product innovation, clinical evidence, and physician education to advance the standard of care for bifurcated coronary artery disease."
The Tryton Side Branch Stent is commercially available in Europe and parts of the Middle East, is investigational in the United States, and is not available in Japan.
Coronary artery disease often results in the buildup of plaque at the site of a bifurcation, where one artery branches from another. Current approaches to treating these lesions are time consuming and technically difficult. As a result, the side branch is often left unstented, leaving it vulnerable to higher rates of restenosis, the re-narrowing of the stented vessel following implantation.
Tryton Medical develops stent systems for the treatment of bifurcation lesions. The company was founded in 2003 in Durham, N.C., by Aaron V. Kaplan, M.D. (professor of medicine at Dartmouth Medical School/Dartmouth-Hitchcock Medical Center) and Dan Cole, general partner at Spray Ventures. Privately held, Tryton is backed by PTV Sciences, RiverVest Venture Partners, Spray Venture Partners, and the 3x5 Special Opportunity Fund.
Tryton Medical develops stent systems for the treatment of bifurcation lesions. The company was founded in 2003 in Durham, N.C., by Aaron V. Kaplan, M.D. (professor of medicine at Dartmouth Medical School/Dartmouth- Hitchcock Medical Center) and Dan Cole, General Partner at Spray Ventures. Privately held, Tryton is backed by PTV Sciences, RiverVest Venture Partners, Spray Venture Partners, and the 3x5 Special Opportunity Fund. - See more at: http://dev.mpo-mag.com/contents/view_breaking-news/2014-02-14/tryton-medical-establishes-bifurcation-institute/#sthash.utYIYNNJ.dpuf