01.28.14
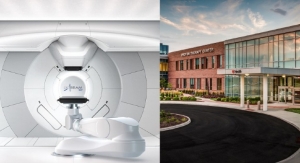
Sumitomo Heavy Industries, Ltd. has received U.S. Food and Drug Administration 510(k) clearance of its Proton Therapy System. Executives said the system is the first 510(k) cleared medical device for Sumitomo.
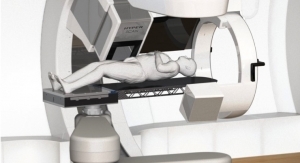
Proton therapy is a form of radiation therapy, which employs proton beams to kill cancer cells with better dose distribution compared with conventional X-rays. Currently, 12 facilities are operational and several more are under construction in the United States, and the U.S. proton therapy market is expected to expand in the future.
Proton beam has a unique characteristic called the “Bragg Peak.” More absorption dose is delivered as the proton loses its energy; consequently, the maximum dose is deposited just before the beam stops, and there is no radiation dose to normal cells behind the target tumor volume. This unique characteristic is especially advantageous for cancers around critical organ structures and pediatric cancers.
Sumitomo is one of the world's first proton system suppliers, having delivered its first commercial system in 1997 to the National Cancer Center Hospital East in Kashiwa, Japan. New projects currently are underway in Taiwan, Korea, and Japan. The System has a 230MeV proton accelerator (cyclotron) and 360 degree single or multiple rotating gantries with a robust design.
“With 510(k) clearance, we have received a ticket for admission to the U.S. market, but this is just the beginning of establishing a long-term relationship with equipment users such as hospitals, doctors and cancer patients. We are hoping the Sumitomo Proton Therapy System will be used to save many cancer patients in the United States,” said Yukio Kumata, Sumitomo's vice president.
Sumitomo Heavy Industries is a technology-oriented Japanese manufacturer of medical and industrial machinery. Its accelerator technology is used in the semiconductor industry and in the nuclear physics research sector as well as in medical applications. The company, which employs 18,000 workers globally and reported annual sales of $6.2 billion in fiscal 2012, is part of the Sumitomo Business Group.
Proton therapy is a form of radiation therapy, which employs proton beams to kill cancer cells with better dose distribution compared with conventional X-rays. Currently, 12 facilities are operational and several more are under construction in the United States, and the U.S. proton therapy market is expected to expand in the future.
Proton beam has a unique characteristic called the “Bragg Peak.” More absorption dose is delivered as the proton loses its energy; consequently, the maximum dose is deposited just before the beam stops, and there is no radiation dose to normal cells behind the target tumor volume. This unique characteristic is especially advantageous for cancers around critical organ structures and pediatric cancers.
Sumitomo is one of the world's first proton system suppliers, having delivered its first commercial system in 1997 to the National Cancer Center Hospital East in Kashiwa, Japan. New projects currently are underway in Taiwan, Korea, and Japan. The System has a 230MeV proton accelerator (cyclotron) and 360 degree single or multiple rotating gantries with a robust design.
“With 510(k) clearance, we have received a ticket for admission to the U.S. market, but this is just the beginning of establishing a long-term relationship with equipment users such as hospitals, doctors and cancer patients. We are hoping the Sumitomo Proton Therapy System will be used to save many cancer patients in the United States,” said Yukio Kumata, Sumitomo's vice president.
Sumitomo Heavy Industries is a technology-oriented Japanese manufacturer of medical and industrial machinery. Its accelerator technology is used in the semiconductor industry and in the nuclear physics research sector as well as in medical applications. The company, which employs 18,000 workers globally and reported annual sales of $6.2 billion in fiscal 2012, is part of the Sumitomo Business Group.