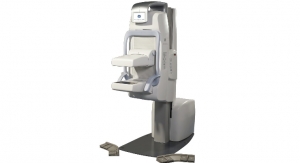
The Invenia Automated Breast Ultrasound System (ABUS) is designed to better detect cancer in women with dense breasts. The system features advanced automation technology and is designed for reproducibility, ease of use and both patient and operator comfort, according to the company. With new tools like compression assist and reverse curve, healthcare providers can capture whole breast, 3-D volumes of clinical images in less time compared with previous versions of the technology (somo•v ABUS).
“The use of ultrasound technology for breast cancer screening is proving to be a valuable asset for healthcare providers in the diagnosis and care provided to patients with dense breasts,” said Anders Wold, president and CEO of GE’s Ultrasound business.
Dense breast tissue is a major risk factor for breast cancer and not only increases the risk of the disease up to four to six times, but also makes cancer more difficult to detect using mammography, according to multiple large studies. One study, published in the New England Journal of Medicine, showed mammography sensitivity is reduced by 36 to 38 percent in women with dense breasts, as density masks the appearance of tumors (Boyd, et al, NEJM 2007:356:227-36M).
To address this limitation with mammography, U-Systems (a GE Healthcare company) introduced the somo•v ABUS in September 2012 to give healthcare providers a U.S. Food and Drug Administration (FDA)-approved screening option for patients with dense breasts.
GE Healthcare provides medical technologies and services to meet the demand for increased access, enhanced quality and more affordable healthcare. The company's products range from medical imaging, software & IT, patient monitoring and diagnostics to drug discovery, biopharmaceutical manufacturing technologies and performance improvement solutions.