11.19.13
The Biomedical division of DSM and Bio2 Medical Inc. are partnering for Bio2's Angel catheter technology.
DSM, a provider of materials and coating technologies for the medical device sector will supply its proprietary ComfortCoat lubricious coating and its coating application expertise.
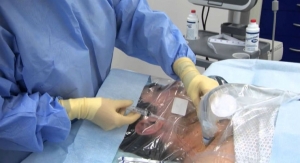
Placed at a patient’s bedside, the Angel catheter is intended to provide inferior vena cava (IVC) filter protection from pulmonary embolism (PE) immediately following admission to the intensive care unit (ICU), while at the same time allowing for central venous access in critically ill patients. PE is a blockage of the main pulmonary artery or one of its branches by a blood clot originating from a different location in the body. A recent report from Millennium Research Group found that as of 2011, there were more than 250,000 cases of confirmed PE treated in U.S. hospitals. That number increases to 344,000 when patients with symptoms consistent with PE are included.
The unique design of the Angel catheter incorporates the PE protection of a retrievable, nitinol IVC filter, permanently attached to a triple-lumen, central venous access catheter. According to Bio2 Medical, the Angel catheter is the first IVC filter to receive CE mark approval for a prophylactic use indication, in addition to traditional IVC filter and central venous catheter indications.
“This partnership allows our flagship product, the Angel catheter, to be easily placed at the patient’s bedside by the attending physician due to DSM’s hydrophilic and lubricous coating technology,” said Jeff Steinmetz, vice president of Research & Development, Bio2 Medical. “We partnered with DSM due to their highly regarded technical support, responsiveness, and ability to meet project timelines.”
“The Angel catheter’s hydrophilic coating makes it easy to place and may reduce vessel site trauma, which is important for my critically ill patients,” said Carl Waldmann, M.D., consultant anesthetist and intensive care physician at Royal Berkshire NHS Trust in Reading, the United Kingdom.
“DSM’s proprietary coating technologies and coating processes havcoatings for DSM Biomedical. “We are proud to see our ComfortCoat coating contributing to the performance of Bio2 Medical’s Angel catheter. The usage in this new catheter not only demonstrates the broad capabilities of our technologies but also underscores how committed DSM and its partners are to addressing unmet market needs with medical innovations.”
The Angel catheter currently is the subject of an FDA early feasibility pilot study. Up to 10 patients will be enrolled in the study with initial results anticipated in the second quarter of 2014.
Netherlands-based DSM serves a variety of industries. Among its offerings, the company's Biomedical division provides biostable polyurethanes; hydrophilic and non-biofouling coatings; polymer stabilizing technology; biomedical polyethylenes; resorbable materials; and silicone hydrogels. The company has 37 locations in the United States across is different divisions.
Bio2 Medical is based in San Antonio, Texas, and has a research and development and manufacturing facility in Golden, Colo.
DSM, a provider of materials and coating technologies for the medical device sector will supply its proprietary ComfortCoat lubricious coating and its coating application expertise.
Placed at a patient’s bedside, the Angel catheter is intended to provide inferior vena cava (IVC) filter protection from pulmonary embolism (PE) immediately following admission to the intensive care unit (ICU), while at the same time allowing for central venous access in critically ill patients. PE is a blockage of the main pulmonary artery or one of its branches by a blood clot originating from a different location in the body. A recent report from Millennium Research Group found that as of 2011, there were more than 250,000 cases of confirmed PE treated in U.S. hospitals. That number increases to 344,000 when patients with symptoms consistent with PE are included.
The unique design of the Angel catheter incorporates the PE protection of a retrievable, nitinol IVC filter, permanently attached to a triple-lumen, central venous access catheter. According to Bio2 Medical, the Angel catheter is the first IVC filter to receive CE mark approval for a prophylactic use indication, in addition to traditional IVC filter and central venous catheter indications.
“This partnership allows our flagship product, the Angel catheter, to be easily placed at the patient’s bedside by the attending physician due to DSM’s hydrophilic and lubricous coating technology,” said Jeff Steinmetz, vice president of Research & Development, Bio2 Medical. “We partnered with DSM due to their highly regarded technical support, responsiveness, and ability to meet project timelines.”
“The Angel catheter’s hydrophilic coating makes it easy to place and may reduce vessel site trauma, which is important for my critically ill patients,” said Carl Waldmann, M.D., consultant anesthetist and intensive care physician at Royal Berkshire NHS Trust in Reading, the United Kingdom.
“DSM’s proprietary coating technologies and coating processes havcoatings for DSM Biomedical. “We are proud to see our ComfortCoat coating contributing to the performance of Bio2 Medical’s Angel catheter. The usage in this new catheter not only demonstrates the broad capabilities of our technologies but also underscores how committed DSM and its partners are to addressing unmet market needs with medical innovations.”
The Angel catheter currently is the subject of an FDA early feasibility pilot study. Up to 10 patients will be enrolled in the study with initial results anticipated in the second quarter of 2014.
Netherlands-based DSM serves a variety of industries. Among its offerings, the company's Biomedical division provides biostable polyurethanes; hydrophilic and non-biofouling coatings; polymer stabilizing technology; biomedical polyethylenes; resorbable materials; and silicone hydrogels. The company has 37 locations in the United States across is different divisions.
Bio2 Medical is based in San Antonio, Texas, and has a research and development and manufacturing facility in Golden, Colo.