10.29.13
Transcatheter Technologies GmbH, is a start-up medical device company that’s developing a next-generation transcatheter aortic heart valve prosthesis. The company is taking on the big boys in what’s becoming an increasingly competitive transcatheter aortic valve implantation (TAVI) sector.
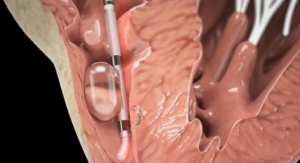
During this week’s annual Transcatheter Cardiovascular Therapeutics (TCT) conference held in San Francisco, Calif., the company reported the first in-human implantation of its transapical Trinity aortic valve.
“A major limitation of existing transcatheter aortic heart valves is that they cannot be repositioned once implanted. Trinity, however, is designed to solve this difficult problem,” said Wolfgang Goetz, M.D., Ph.D., CEO, and a cardiac surgeon by training. “With Trinity, once our valve is initially placed, a cardiologist can fully evaluate the valve’s function to determine whether it needs to be repositioned, retrieved or kept in the same position. Equally important, Trinity provides for a controlled positioning by allowing a cardiologist to slowly open the valve stent, thus improving the likelihood of a first-attempt correct placement.”
“The Trinity aortic valve can be positioned precisely. Yet, it also allows for safe repositioning of the valve prosthesis—or even full retrieval—if necessary. Its unique sealing cuff provides excellent results without paravalvular leakage. Moreover, the risk for atrio-ventricular block is dramatically reduced due to the supra-annular positioning of Trinity,” added Christian Hengstenberg, M.D., cardiologist at the German Heart Center in Munich.
Transcatheter Technologies is a privately held medical device company founded in 2009 and headquartered in Regensburg, Germany.
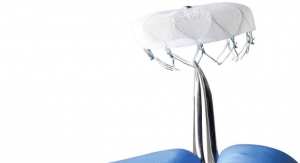
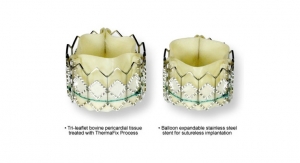
The Trinity aortic valve prosthesis is comprises a bovine pericardium valve with porcine pericardium-sealing cuff that is mounted on a self-expanding nitinol frame and pre-mounted on a detachable catheter tip.
But the company is vying for position in a market populated with heavy-hitters such as Medtronic Inc., Edwards Lifesciences Corp., St. Jude Medical Inc., and a slew of start-ups and midsize companies bucking for position.
Earlier this month, Medtronic launched a 60-patient clinical trial of its CoreValve Evolut R repositionable heart valve, consisting of the CoreValve Evolut R device and its EnVeo R delivery system, which the company bills as “recapturable.” In November last year, St. Jude Medical got CE mark approval in the European Union for its repositionable and retrievable Portico TAVI system.
During this week’s annual Transcatheter Cardiovascular Therapeutics (TCT) conference held in San Francisco, Calif., the company reported the first in-human implantation of its transapical Trinity aortic valve.
“A major limitation of existing transcatheter aortic heart valves is that they cannot be repositioned once implanted. Trinity, however, is designed to solve this difficult problem,” said Wolfgang Goetz, M.D., Ph.D., CEO, and a cardiac surgeon by training. “With Trinity, once our valve is initially placed, a cardiologist can fully evaluate the valve’s function to determine whether it needs to be repositioned, retrieved or kept in the same position. Equally important, Trinity provides for a controlled positioning by allowing a cardiologist to slowly open the valve stent, thus improving the likelihood of a first-attempt correct placement.”
“The Trinity aortic valve can be positioned precisely. Yet, it also allows for safe repositioning of the valve prosthesis—or even full retrieval—if necessary. Its unique sealing cuff provides excellent results without paravalvular leakage. Moreover, the risk for atrio-ventricular block is dramatically reduced due to the supra-annular positioning of Trinity,” added Christian Hengstenberg, M.D., cardiologist at the German Heart Center in Munich.
Transcatheter Technologies is a privately held medical device company founded in 2009 and headquartered in Regensburg, Germany.
The Trinity aortic valve prosthesis is comprises a bovine pericardium valve with porcine pericardium-sealing cuff that is mounted on a self-expanding nitinol frame and pre-mounted on a detachable catheter tip.
But the company is vying for position in a market populated with heavy-hitters such as Medtronic Inc., Edwards Lifesciences Corp., St. Jude Medical Inc., and a slew of start-ups and midsize companies bucking for position.
Earlier this month, Medtronic launched a 60-patient clinical trial of its CoreValve Evolut R repositionable heart valve, consisting of the CoreValve Evolut R device and its EnVeo R delivery system, which the company bills as “recapturable.” In November last year, St. Jude Medical got CE mark approval in the European Union for its repositionable and retrievable Portico TAVI system.