10.02.13
Russ Trenary has been named president and CEO of Miami, Fla.-based InnFocus Inc., a privately held medical device company with biomaterials expertise that has developed a patented micro-shunt for treating glaucoma. The company's main product, called the InnFocus MicroShunt, is used in minimally-invasive glaucoma surgery outside of the United States.
Company founder, Leonard Pinchuk, Ph.D., will remain with InnFocus to develop its product pipeline.
The device is cleared by the U.S. Food and Drug Administration (FDA) to begin Phase I clinical trials. The company has raised $13.4 million so far in a Series B financing led by the Hoya Group, a Japan-based manufacturer of ophthalmology products. InnFocus is in the process of finishing the Series B offering to raise an additional $3 million. The funds will be used to complete the Phase I clinical trials, expand clinical studies in Europe where the device is CE mark approved, and to invest in growth initiatives.
Trenary has spent more than 30 years with ophthalmic medical device companies, including Allergan Inc. and Advanced Medical Optics. His achievements include the launch of many successful ophthalmic devices.
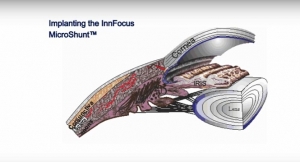
The InnFocus MicroShunt (formerly known as the MIDI Arrow) is a glaucoma drainage implant consisting of a micro-tube about twice the size of an eyelash with "fins" located halfway down its length. It is made from a proprietary biomaterial that, according to the company, "demonstrates insignificant inflammation and encapsulation in the eye." The device can be implanted alone or in combination with cataract surgery. The technology was developed in collaboration with the University of Miami, Miller School of Medicine, Bascom Palmer Eye Institute.
"The scientific team at InnFocus is very impressive, and I believe that the sustained IOP (intraocular pressure—the fluid pressure inside the eye) reductions with the vast majority of pressures being below 14 mmHg (millimeters of mercury) represent ground breaking results," said Trenary. "These pressures in the low teens differentiate the InnFocus MicroShunt from the other glaucoma devices currently in clinical trials (known as the "MIGS" devices) that tend to achieve more modest IOP reductions. We are eager to complete the Phase I FDA trials, and continue work outside of the United States to establish the InnFocus MicroShunt as the premier offering in the minimally invasive glaucoma arena."
InnFocus has been testing several generations of its glaucoma shunt in France and the Dominican Republic for more than six years. In an ongoing study, now in its third year in the Dominican Republic, the company has reported that the InnFocus MicroShunt can lower intraocular pressure by 50-60 percent with the average pressure residing between 10 and 12 mmHg. Scientific studies have shown that reducing pressure below 14 mmHg can arrest the progression of the glaucoma. According to the company's data, 90 percent of the eyes that have received the MicroShunt have IOPs less than 14 mmHg with no long-term adverse events and with approximately 90 percent of the patients completely off of glaucoma medication. One of the keys to success is the biomaterial used in the device that "demonstrates a clinically insignificant foreign body reaction in the eye," company officials report.
"We are at the perfect juncture in our company's history to bring in an accomplished, experienced veteran like Russ Trenary," said Pinchuk, "His demonstrated leadership in developing and launching ophthalmic devices and his broad network of relationships with leading eye surgeons are a perfect blend. We look forward to having him guide us through the FDA and overseas clinical processes."
Company founder, Leonard Pinchuk, Ph.D., will remain with InnFocus to develop its product pipeline.
The device is cleared by the U.S. Food and Drug Administration (FDA) to begin Phase I clinical trials. The company has raised $13.4 million so far in a Series B financing led by the Hoya Group, a Japan-based manufacturer of ophthalmology products. InnFocus is in the process of finishing the Series B offering to raise an additional $3 million. The funds will be used to complete the Phase I clinical trials, expand clinical studies in Europe where the device is CE mark approved, and to invest in growth initiatives.
Trenary has spent more than 30 years with ophthalmic medical device companies, including Allergan Inc. and Advanced Medical Optics. His achievements include the launch of many successful ophthalmic devices.
The InnFocus MicroShunt (formerly known as the MIDI Arrow) is a glaucoma drainage implant consisting of a micro-tube about twice the size of an eyelash with "fins" located halfway down its length. It is made from a proprietary biomaterial that, according to the company, "demonstrates insignificant inflammation and encapsulation in the eye." The device can be implanted alone or in combination with cataract surgery. The technology was developed in collaboration with the University of Miami, Miller School of Medicine, Bascom Palmer Eye Institute.
"The scientific team at InnFocus is very impressive, and I believe that the sustained IOP (intraocular pressure—the fluid pressure inside the eye) reductions with the vast majority of pressures being below 14 mmHg (millimeters of mercury) represent ground breaking results," said Trenary. "These pressures in the low teens differentiate the InnFocus MicroShunt from the other glaucoma devices currently in clinical trials (known as the "MIGS" devices) that tend to achieve more modest IOP reductions. We are eager to complete the Phase I FDA trials, and continue work outside of the United States to establish the InnFocus MicroShunt as the premier offering in the minimally invasive glaucoma arena."
InnFocus has been testing several generations of its glaucoma shunt in France and the Dominican Republic for more than six years. In an ongoing study, now in its third year in the Dominican Republic, the company has reported that the InnFocus MicroShunt can lower intraocular pressure by 50-60 percent with the average pressure residing between 10 and 12 mmHg. Scientific studies have shown that reducing pressure below 14 mmHg can arrest the progression of the glaucoma. According to the company's data, 90 percent of the eyes that have received the MicroShunt have IOPs less than 14 mmHg with no long-term adverse events and with approximately 90 percent of the patients completely off of glaucoma medication. One of the keys to success is the biomaterial used in the device that "demonstrates a clinically insignificant foreign body reaction in the eye," company officials report.
"We are at the perfect juncture in our company's history to bring in an accomplished, experienced veteran like Russ Trenary," said Pinchuk, "His demonstrated leadership in developing and launching ophthalmic devices and his broad network of relationships with leading eye surgeons are a perfect blend. We look forward to having him guide us through the FDA and overseas clinical processes."