07.03.13
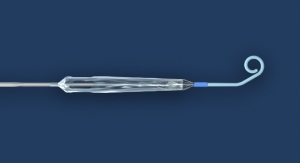
Pleasanton, Calif.-based Thoratec Corp. has acquired the DuraHeart II (DH-II) ventricular assist system from Terumo Corporation for $13 million in cash and potential future milestone payments, based on regulatory approvals and product sales, of up to $43.5 million.
As part of the agreement, a team of Terumo employees will transition to Thoratec and will continue to be based in Ann Arbor, Michigan, where Terumo Cardiovascular Systems is based. Thoratec and Terumo also formed distribution partnership in which Terumo will sell the system in Japan and potentially other parts of Asia.
A ventricular assist device (VAD) is used to partially or completely to replace the function of a failing heart. According to Thoratec, the system is an “ultra-compact, full-support, centrifugal flow chronic VAD that uses a unique technology foundation known as ‘force balance’ suspension.” The system uses primary magnetic forces, balanced by hydrodynamic support, to achieve consistent gaps across the operating range of the pump, independent of pump speed. This approach is designed to create a pumping mechanism with excellent blood-handling characteristics, according to Terumo and Thoratec. Preclinical testing has shown a favorable profile for DH-II with respect to hemolysis, bleeding and thrombosis, the companies reported.
Thoratec plans to use its expertise in mechanical circulatory support in order to advance the DH-II program through product development and clinical trials. Thoratec anticipates a first-in-human implant of the DH-II pump in 2016 to be followed by clinical trials in the United States and abroad.
"Thoratec is committed to delivering a steady cadence of innovative new products to the VAD market in the coming years," said Gary F. Burbach, president and CEO of Thoratec. "DuraHeart II brings a differentiated approach to mechanical circulatory support to Thoratec's R&D portfolio and will be an integral component of our product development strategy, along with continued evolution of the HeartMate II system, next-generation pump platforms including HeartMate IIIT and HeartMate PHP, which are expected to begin pivotal CE mark trials in the second half of 2013, and breakthrough cross-platform technologies such as our fully implantable system."
Thoratec anticipates that the acquisition will add incremental ongoing operating expenses, primarily related to research and development, of approximately $6 million to $7 million in the second half of 2013. Additionally, Thoratec will recognize transaction-related expenses, including the amortization of intangible assets, which will be quantified in the company's second quarter earnings report and following the completion of the purchase price allocation for the transaction.
Thoratec develops therapies to address advanced-stage heart failure.
Tokyo, Japan-based Terumo Corporation develops and manufactures devices for use in cardiothoracic surgery, interventional procedures, and transfusion medicine. The company also manufactures a broad array of syringe and hypodermic needle products for hospital and physician office use. Terumo Medical’s U.S. headquarters are in Somerset, N.J.
As part of the agreement, a team of Terumo employees will transition to Thoratec and will continue to be based in Ann Arbor, Michigan, where Terumo Cardiovascular Systems is based. Thoratec and Terumo also formed distribution partnership in which Terumo will sell the system in Japan and potentially other parts of Asia.
A ventricular assist device (VAD) is used to partially or completely to replace the function of a failing heart. According to Thoratec, the system is an “ultra-compact, full-support, centrifugal flow chronic VAD that uses a unique technology foundation known as ‘force balance’ suspension.” The system uses primary magnetic forces, balanced by hydrodynamic support, to achieve consistent gaps across the operating range of the pump, independent of pump speed. This approach is designed to create a pumping mechanism with excellent blood-handling characteristics, according to Terumo and Thoratec. Preclinical testing has shown a favorable profile for DH-II with respect to hemolysis, bleeding and thrombosis, the companies reported.
Thoratec plans to use its expertise in mechanical circulatory support in order to advance the DH-II program through product development and clinical trials. Thoratec anticipates a first-in-human implant of the DH-II pump in 2016 to be followed by clinical trials in the United States and abroad.
"Thoratec is committed to delivering a steady cadence of innovative new products to the VAD market in the coming years," said Gary F. Burbach, president and CEO of Thoratec. "DuraHeart II brings a differentiated approach to mechanical circulatory support to Thoratec's R&D portfolio and will be an integral component of our product development strategy, along with continued evolution of the HeartMate II system, next-generation pump platforms including HeartMate IIIT and HeartMate PHP, which are expected to begin pivotal CE mark trials in the second half of 2013, and breakthrough cross-platform technologies such as our fully implantable system."
Thoratec anticipates that the acquisition will add incremental ongoing operating expenses, primarily related to research and development, of approximately $6 million to $7 million in the second half of 2013. Additionally, Thoratec will recognize transaction-related expenses, including the amortization of intangible assets, which will be quantified in the company's second quarter earnings report and following the completion of the purchase price allocation for the transaction.
Thoratec develops therapies to address advanced-stage heart failure.
Tokyo, Japan-based Terumo Corporation develops and manufactures devices for use in cardiothoracic surgery, interventional procedures, and transfusion medicine. The company also manufactures a broad array of syringe and hypodermic needle products for hospital and physician office use. Terumo Medical’s U.S. headquarters are in Somerset, N.J.