Niki Arrowsmith05.30.13
Minneapolis, Minn.-based medical device powerhouse Medtronic Inc. is expanding its portfolio of endovascular aortic repair products in the United States with two new medical devices. The company recently received approval from the U.S. Food and Drug Administration (FDA) for the Endurant II Aorto-Uni-Iliac (AUI) stent graft system and FDA 510(k) clearance for the Sentrant introducer sheath.
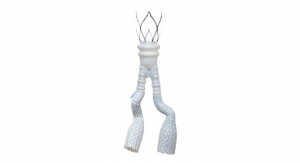
According to Medtronic, the Endurant II AUI stent graft system is the only FDA-approved AUI device in the United States indicated for the primary endovascular treatment of infrarenal abdominal aortic or aorto-iliac aneurysms in patients whose anatomy does not allow for the use of a bifurcated device. Both the bifurcated and AUI configurations of the Endurant stent graft system provide a new pathway for blood flow through the iliac arteries in abdominal aortic aneurysms, thereby reducing risk of aneurysm rupture.
While use of the bifurcated device requires access to both iliac arteries, the AUI device requires access to only one iliac artery. In published studies of endovascular abdominal aortic aneurysm (AAA) repair, current global usage of AUI stent graft configurations averages 5 percent (range 0 to 26 percent) for intact AAA and 39 percent (range 0 to 91 percent) for ruptured AAA.
“The new Endurant II Aorto-Uni-Iliac stent graft extends the performance of the Endurant system to patients with difficult access,” said Michel Makaroun, M.D., chief of vascular surgery at the University of Pittsburgh Medical Center in Pennsylvania and co-director of the university’s Heart and Vascular Institute. “By maintaining the deliverability, conformability and deployment accuracy of the bifurcated Endurant device, the AUI configuration offers aneurysm patients with challenging outflow anatomies a better option for a successful endovascular aortic repair.”
As with the bifurcated Endurant II stent graft, distinguishing features of the Endurant II AUI stent graft include a low delivery profile, tip capture for easy and accurate deployment and compatibility with contralateral iliac limbs and aortic extensions for ultimate patient applicability.
The Sentrant introducer sheath is inserted at the access site in the patient’s femoral artery and advanced upwards into the iliac arteries to facilitate the implant procedure and enable smooth passage of the stent graft delivery system en route to the treatment site in the aorta.
The Sentrant introducer sheath comes in diameters of 12-26 French and shaft lengths of 28 cm. The accessory device also has a reinforced coil for kink resistance, hydrophilic coating and flexibility to improve tracking through tortuous and calcified iliacs and a dilator locking mechanism for secure positioning.
The sheath received the CE Mark in April 2013.
According to Medtronic, the Endurant II AUI stent graft system is the only FDA-approved AUI device in the United States indicated for the primary endovascular treatment of infrarenal abdominal aortic or aorto-iliac aneurysms in patients whose anatomy does not allow for the use of a bifurcated device. Both the bifurcated and AUI configurations of the Endurant stent graft system provide a new pathway for blood flow through the iliac arteries in abdominal aortic aneurysms, thereby reducing risk of aneurysm rupture.
While use of the bifurcated device requires access to both iliac arteries, the AUI device requires access to only one iliac artery. In published studies of endovascular abdominal aortic aneurysm (AAA) repair, current global usage of AUI stent graft configurations averages 5 percent (range 0 to 26 percent) for intact AAA and 39 percent (range 0 to 91 percent) for ruptured AAA.
“The new Endurant II Aorto-Uni-Iliac stent graft extends the performance of the Endurant system to patients with difficult access,” said Michel Makaroun, M.D., chief of vascular surgery at the University of Pittsburgh Medical Center in Pennsylvania and co-director of the university’s Heart and Vascular Institute. “By maintaining the deliverability, conformability and deployment accuracy of the bifurcated Endurant device, the AUI configuration offers aneurysm patients with challenging outflow anatomies a better option for a successful endovascular aortic repair.”
As with the bifurcated Endurant II stent graft, distinguishing features of the Endurant II AUI stent graft include a low delivery profile, tip capture for easy and accurate deployment and compatibility with contralateral iliac limbs and aortic extensions for ultimate patient applicability.
The Sentrant introducer sheath is inserted at the access site in the patient’s femoral artery and advanced upwards into the iliac arteries to facilitate the implant procedure and enable smooth passage of the stent graft delivery system en route to the treatment site in the aorta.
The Sentrant introducer sheath comes in diameters of 12-26 French and shaft lengths of 28 cm. The accessory device also has a reinforced coil for kink resistance, hydrophilic coating and flexibility to improve tracking through tortuous and calcified iliacs and a dilator locking mechanism for secure positioning.
The sheath received the CE Mark in April 2013.